Predominant formation of heavily pigmented dermal melanocytomas resembling 'animal-type' melanomas in hepatocyte growth factor (C57BL/6 x C3H)F1 mice following neonatal UV irradiation
- PMID: 17696912
- PMCID: PMC2410209
- DOI: 10.1111/j.1600-0560.2006.00679.x
Predominant formation of heavily pigmented dermal melanocytomas resembling 'animal-type' melanomas in hepatocyte growth factor (C57BL/6 x C3H)F1 mice following neonatal UV irradiation
Abstract
Background: Transgenic mice expressing hepatocyte growth factor (HGF) develop cutaneous melanocytic tumors following neonatal UV exposure. Here, we examined the histologic spectrum of UV-induced melanocytic tumors in HGF mice on a pigmented (C57BL/6 x C3H/HeN)F(1) background.
Methods: Neonatally irradiated (4000 J/m(2)) mice were monitored for 43 weeks, and 31/34 (91%) animals developed a total of 163 melanocytic tumors.
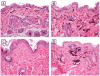
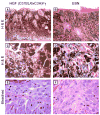
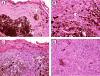
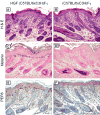
Results: Of 54 primary tumors analyzed, most (49/54, 91%) demonstrated exclusively dermal collections of epithelioid cells with voluminous densely pigmented cytoplasm. Seven of these also demonstrated a population of spindled cells with mitoses. Several (3/54, 6%) tumors exhibited a junctional component with melanocytes present in the epidermis. Staining with PEP8 confirmed the presence of interfollicular melanocytes at the dermal-epidermal junction in neonatal skin.
Conclusions: In contrast to HGF animals on an albino (FVB) background, HGF animals on the pigmented (C57BL/6 x C3H/HeN)F(1) background do not develop classic radial growth phase melanoma but rather predominantly develop dermal melanocytomas resembling the 'animal-type' melanoma occasionally seen in humans. These results demonstrate the influence of genetic background on histologic pattern of UV-induced melanomas in mice.
Figures
Similar articles
-
Accelerated ultraviolet radiation-induced carcinogenesis in hepatocyte growth factor/scatter factor transgenic mice.Cancer Res. 2000 Jul 15;60(14):3738-43. Cancer Res. 2000. PMID: 10919643
-
Neonatal susceptibility to UV induced cutaneous malignant melanoma in a mouse model.Photochem Photobiol Sci. 2006 Feb;5(2):254-60. doi: 10.1039/b506974b. Epub 2005 Jul 27. Photochem Photobiol Sci. 2006. PMID: 16465311 Review.
-
Melanocyte expression of survivin promotes development and metastasis of UV-induced melanoma in HGF-transgenic mice.Cancer Res. 2007 Jun 1;67(11):5172-8. doi: 10.1158/0008-5472.CAN-06-3669. Cancer Res. 2007. PMID: 17545596 Free PMC article.
-
Neonatal UVB exposure accelerates melanoma growth and enhances distant metastases in Hgf-Cdk4(R24C) C57BL/6 mice.Int J Cancer. 2011 Jul 15;129(2):285-94. doi: 10.1002/ijc.25913. Epub 2011 Apr 21. Int J Cancer. 2011. PMID: 21207411
-
UV-Induced Molecular Signaling Differences in Melanoma and Non-melanoma Skin Cancer.Adv Exp Med Biol. 2017;996:27-40. doi: 10.1007/978-3-319-56017-5_3. Adv Exp Med Biol. 2017. PMID: 29124688 Review.
Cited by
-
A murine model for the development of melanocytic nevi and their progression to melanoma.Mol Carcinog. 2016 May;55(5):646-58. doi: 10.1002/mc.22310. Epub 2015 Mar 18. Mol Carcinog. 2016. PMID: 25788145 Free PMC article.
-
Superficial spreading-like melanoma in Arf(-/-)::Tyr-Nras(Q61K)::K14-Kitl mice: keratinocyte Kit ligand expression sufficient to "translocate" melanomas from dermis to epidermis.J Invest Dermatol. 2011 Jun;131(6):1384-7. doi: 10.1038/jid.2011.21. Epub 2011 Feb 10. J Invest Dermatol. 2011. PMID: 21307875 Free PMC article. No abstract available.
-
NM23 deficiency promotes metastasis in a UV radiation-induced mouse model of human melanoma.Clin Exp Metastasis. 2013 Jan;30(1):25-36. doi: 10.1007/s10585-012-9495-z. Epub 2012 Jun 15. Clin Exp Metastasis. 2013. PMID: 22699362 Free PMC article.
-
N-acetylcysteine protects melanocytes against oxidative stress/damage and delays onset of ultraviolet-induced melanoma in mice.Clin Cancer Res. 2007 Oct 1;13(19):5952-8. doi: 10.1158/1078-0432.CCR-07-1187. Clin Cancer Res. 2007. PMID: 17908992 Free PMC article.
References
-
- Berkelhammer J, Oxenhandler RW, Hook RR, Jr, Hennessy JM. Development of a new melanoma model in C57BL/6 mice. Cancer Res. 1982;42:3157–3163. - PubMed
-
- Broome Powell M, Gause PR, Hyman P, Gregus J, Lluria-Prevatt M, Nagle R, Bowden GT. Induction of melanoma in TPras transgenic mice. Carcinogenesis. 1999;20:1747–1453. - PubMed
-
- Carney JA, Ferreiro JA. The epithelioid blue nevus. A multicentric familial tumor with important associations, including cardiac myxoma and psammomatous melanotic schwannoma. Am J Surg Pathol. 1996;20:259–272. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

