The bundlin pilin protein of enteropathogenic Escherichia coli is an N-acetyllactosamine-specific lectin
- PMID: 17697132
- PMCID: PMC3809902
- DOI: 10.1111/j.1462-5822.2007.01028.x
The bundlin pilin protein of enteropathogenic Escherichia coli is an N-acetyllactosamine-specific lectin
Abstract
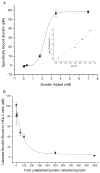
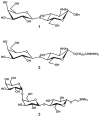
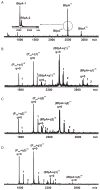
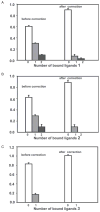
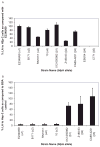
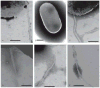
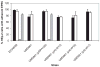
Synthetic N-acetyllactosamine (LacNAc) glycoside sequences coupled to BSA competitively inhibit enteropathogenic Escherichia coli (EPEC) localized adherence (LA) to human intestinal biopsy specimens and tissue culture cell monolayers. The LacNAc-specific adhesin appears to be associated with the bundle-forming pili (BFP) expressed by EPEC during the early stages of colonization. Herein, we report that recombinant bundlin inhibits EPEC LA to HEp-2 cells and binds to HEp-2 cells. Recombinant bundlin also binds, with millimolar association constants (K(assoc)), to synthetic LacNAc-Benzene and LacNAc-O(CH(2))(8)CONH(2) glycosides as assessed in the gas phase by nanoelectrospray ionization mass spectrometry. Furthermore, LacNAc-BSA inhibits LA only of EPEC strains that express alpha bundlin alleles, suggesting putative locations for the LacNAc-binding pocket in the alpha bundlin monomer. Collectively, these results suggest that alpha bundlin possesses lectin-like properties that are responsible for LacNAc-specific initial adherence of alpha bundlin-expressing EPEC strains to host intestinal epithelial cells.
Figures

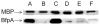
References
-
- Armstrong GD, Mulvey GL, Marcato P, Griener TP, Kahan MC, Tennent GA, et al. Human serum amyloid P component protects against Escherichia coli O157:H7 Shiga toxin 2 in vivo: therapeutic implications for hemolytic-uremic syndrome. J Infect Dis. 2006;193:1120–1124. - PubMed
-
- Barnett Foster D, Philpott D, Abul-Milh M, Huesca M, Sherman PM, Lingwood CA. Phosphatidylethanolamine recognition promotes enteropathogenic E. coli and enterohemorrhagic E. coli host cell attachment. Microb Pathog. 1999;27:289–301. - PubMed
-
- Bieber D, Ramer SW, Wu CY, Murray WJ, Tobe T, Fernandez R, Schoolnik GK. Type IV pili, transient bacterial aggregates, and virulence of enteropathogenic Escherichia coli. Science. 1998;280:2114–2118. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

