Differential ex vivo nitric oxide production by acutely isolated neonatal and adult microglia
- PMID: 17698208
- PMCID: PMC2040026
- DOI: 10.1016/j.jneuroim.2007.07.004
Differential ex vivo nitric oxide production by acutely isolated neonatal and adult microglia
Abstract
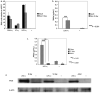
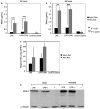
Microglia are the macrophage population residing in the parenchyma of the central nervous system (CNS), and are thought to play critical roles in CNS development, homeostasis and defense against pathogens. Microglia are capable of rapidly responding to microbial pathogens through engagement of their Toll-like receptors (TLRs). We first compared the efficiency of these responses in primary microglia acutely isolated from adult and neonatal mice. While the cytokine and chemokine responses of adult microglia were generally higher than those of neonatal cells stimulated ex vivo through TLRs, the nitric oxide response of neonatal microglia was markedly enhanced relative to the adult cells. We then went on to identify culture conditions such as exposure to M-SCF or GM-CSF that markedly enhanced the nitric oxide response of microglia, particularly those from the adult CNS. Finally, we demonstrate that the differential nitric oxide response of neonatal and adult microglia is not only limited to the mouse, but also extends to rat microglia.
Figures





References
-
- Akagawa KS. Functional heterogeneity of colony-stimulating factor-induced human monocyte-derived macrophages. Int J Hematol. 2002;76:27–34. - PubMed
-
- Albina JE, Abate JA, Henry WL., Jr. Nitric oxide production is required for murine resident peritoneal macrophages to suppress mitogen-stimulated T cell proliferation. Role of IFN-gamma in the induction of the nitric oxide-synthesizing pathway. J Immunol. 1991;147:144–148. - PubMed
-
- Brannan CA, Roberts MR. Resident microglia from adult mice are refractory to nitric oxide-inducing stimuli due to impaired NOS2 gene expression. Glia. 2004;48:120–131. - PubMed
-
- Butovsky O, Ziv Y, Schwartz A, Landa G, Talpalar AE, Pluchino S, Martino G, Schwartz M. Microglia activated by IL-4 or IFN-gamma differentially induce neurogenesis and oligodendrogenesis from adult stem/progenitor cells. Mol Cell Neurosci. 2006;31:149–160. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

