Taenia solium oncosphere adhesion to intestinal epithelial and Chinese hamster ovary cells in vitro
- PMID: 17698575
- PMCID: PMC2168301
- DOI: 10.1128/IAI.01175-06
Taenia solium oncosphere adhesion to intestinal epithelial and Chinese hamster ovary cells in vitro
Abstract
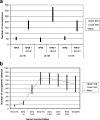
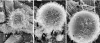
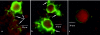
The specific mechanisms underlying Taenia solium oncosphere adherence and penetration in the host have not been studied previously. We developed an in vitro adhesion model assay to evaluate the mechanisms of T. solium oncosphere adherence to the host cells. The following substrates were used: porcine intestinal mucosal scrapings (PIMS), porcine small intestinal mucosal explants (PSIME), Chinese hamster ovary cells (CHO cells), epithelial cells from ileocecal colorectal adenocarcinoma (HCT-8 cells), and epithelial cells from colorectal adenocarcinoma (Caco-2 cells). CHO cells were used to compare oncosphere adherence to fixed and viable cells, to determine the optimum time of oncosphere incubation, to determine the role of sera and monolayer cell maturation, and to determine the effect of temperature on oncosphere adherence. Light microscopy, scanning microscopy, and transmission microscopy were used to observe morphological characteristics of adhered oncospheres. This study showed in vitro adherence of activated T. solium oncospheres to PIMS, PSIME, monolayer CHO cells, Caco-2 cells, and HCT-8 cells. The reproducibility of T. solium oncosphere adherence was most easily measured with CHO cells. Adherence was enhanced by serum-binding medium with >5% fetal bovine serum, which resulted in a significantly greater number of oncospheres adhering than the number adhering when serum at a concentration less than 2.5% was used (P < 0.05). Oncosphere adherence decreased with incubation of cells at 4 degrees C compared with the adherence at 37 degrees C. Our studies also demonstrated that T. solium oncospheres attach to cells with elongated microvillus processes and that the oncospheres expel external secretory vesicles that have the same oncosphere processes.
Figures




Similar articles
-
Standardization of a fluorescent-based quantitative adhesion assay to study attachment of Taenia solium oncosphere to epithelial cells in vitro.J Immunol Methods. 2012 Feb 28;376(1-2):89-96. doi: 10.1016/j.jim.2011.12.001. Epub 2011 Dec 9. J Immunol Methods. 2012. PMID: 22178422 Free PMC article.
-
[Observation on the ultrastructure of Taenia solium oncosphere].Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2009 Feb 28;27(1):35-8. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2009. PMID: 19459497 Chinese.
-
In vitro oncosphere-killing assays to determine immunity to the larvae of Taenia pisiformis, Taenia ovis, Taenia saginata, and Taenia solium.J Parasitol. 2006 Apr;92(2):273-81. doi: 10.1645/GE-619R.1. J Parasitol. 2006. PMID: 16729683
-
Transmission dynamics of Taenia solium and potential for pig-to-pig transmission.Parasitol Int. 2006;55 Suppl:S131-5. doi: 10.1016/j.parint.2005.11.021. Epub 2005 Dec 13. Parasitol Int. 2006. PMID: 16352459 Review.
-
Vaccination for the prevention of cysticercosis.Dev Biol (Basel). 2004;119:361-8. Dev Biol (Basel). 2004. PMID: 15742647 Review.
Cited by
-
First ultrastructural data on the human tapeworm Taenia asiatica eggs by scanning and transmission electron microscopy (SEM, TEM).Parasitol Res. 2016 Sep;115(9):3649-55. doi: 10.1007/s00436-016-5165-4. Epub 2016 Jun 8. Parasitol Res. 2016. PMID: 27277232
-
Blood-brain barrier disruption and angiogenesis in a rat model for neurocysticercosis.J Neurosci Res. 2019 Feb;97(2):137-148. doi: 10.1002/jnr.24335. Epub 2018 Oct 13. J Neurosci Res. 2019. PMID: 30315659 Free PMC article.
-
Axonal swellings and spheroids: a new insight into the pathology of neurocysticercosis.Brain Pathol. 2019 May;29(3):425-436. doi: 10.1111/bpa.12669. Epub 2018 Dec 11. Brain Pathol. 2019. PMID: 30368965 Free PMC article.
-
Novel rat model for neurocysticercosis using Taenia solium.Am J Pathol. 2015 Aug;185(8):2259-68. doi: 10.1016/j.ajpath.2015.04.015. Am J Pathol. 2015. PMID: 26216286 Free PMC article.
-
Cysticercosis with an Orbital Tropism in Twins.Am J Trop Med Hyg. 2015 Oct;93(4):828-30. doi: 10.4269/ajtmh.15-0205. Epub 2015 Jul 27. Am J Trop Med Hyg. 2015. PMID: 26217041 Free PMC article.
References
-
- Acha, P., and B. Cifres. 1986. Zoonosis y enfermedades transmisibles comunes al hombre y los animals, 2nd ed., p. 763-774. Pan American Health Organization, Washington, DC.
-
- Aspholm-Hurtig, M., G. Dailide, M. Lahmann, A. Kalia, D. Ilver, N. Roche, S. Vikstrom, R. Sjostrom, S. Linden, A. Bakstrom, et al. 2004. Functional adaptation of BabA, the H. pylori ABO blood group antigen binding adhesion. Science 305:519-522. - PubMed
-
- Bilqees, F. M. 1968. Observations on the structure of oncosphere of Taenia crassiceps (Zeder, 1800) Rudolphi, 1810. Can. J. Zool. 46:763-765. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

