Nuclear localization of the ERK MAP kinase mediated by Drosophila alphaPS2betaPS integrin and importin-7
- PMID: 17699602
- PMCID: PMC1995703
- DOI: 10.1091/mbc.e06-07-0659
Nuclear localization of the ERK MAP kinase mediated by Drosophila alphaPS2betaPS integrin and importin-7
Abstract
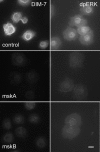
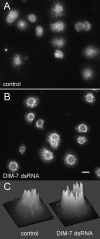
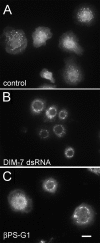
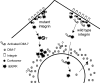
The control of gene expression by the mitogen-activated protein (MAP) kinase extracellular signal-regulated kinase (ERK) requires its translocation into the nucleus. In Drosophila S2 cells nuclear accumulation of diphospho-ERK (dpERK) is greatly reduced by interfering double-stranded RNA against Drosophila importin-7 (DIM-7) or by the expression of integrin mutants, either during active cell spreading or after stimulation by insulin. In both cases, total ERK phosphorylation (on Westerns) is not significantly affected, and ERK accumulates in a perinuclear ring. Tyrosine phosphorylation of DIM-7 is reduced in cells expressing integrin mutants, indicating a mechanistic link between these components. DIM-7 and integrins localize to the same actin-containing peripheral regions in spreading cells, but DIM-7 is not concentrated in paxillin-positive focal contacts or stable focal adhesions. The Corkscrew (SHP-2) tyrosine phosphatase binds DIM-7, and Corkscrew is required for the cortical localization of DIM-7. These data suggest a model in which ERK phosphorylation must be spatially coupled to integrin-mediated DIM-7 activation to make a complex that can be imported efficiently. Moreover, dpERK nuclear import can be restored in DIM-7-deficient cells by Xenopus Importin-7, demonstrating that ERK import is an evolutionarily conserved function of this protein.
Figures


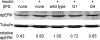

Similar articles
-
Nuclear import of activated D-ERK by DIM-7, an importin family member encoded by the gene moleskin.Development. 2001 Apr;128(8):1403-14. doi: 10.1242/dev.128.8.1403. Development. 2001. PMID: 11262240
-
The Drosophila protein kinase LK6 is regulated by ERK and phosphorylates the eukaryotic initiation factor eIF4E in vivo.Biochem J. 2005 Feb 1;385(Pt 3):695-702. doi: 10.1042/BJ20040769. Biochem J. 2005. PMID: 15487973 Free PMC article.
-
Tyrosine phosphatase SHP-2 regulates IL-1 signaling in fibroblasts through focal adhesions.J Cell Physiol. 2006 Apr;207(1):132-43. doi: 10.1002/jcp.20544. J Cell Physiol. 2006. PMID: 16250012
-
Identification and characterization of Drosophila Snurportin reveals a role for the import receptor Moleskin/importin-7 in snRNP biogenesis.Mol Biol Cell. 2013 Sep;24(18):2932-42. doi: 10.1091/mbc.E13-03-0118. Epub 2013 Jul 24. Mol Biol Cell. 2013. PMID: 23885126 Free PMC article.
-
ERK signaling dynamics in the morphogenesis and homeostasis of Drosophila.Curr Opin Genet Dev. 2020 Aug;63:9-15. doi: 10.1016/j.gde.2020.01.004. Epub 2020 Mar 4. Curr Opin Genet Dev. 2020. PMID: 32145545 Review.
Cited by
-
Dystrophin- and Utrophin-Based Therapeutic Approaches for Treatment of Duchenne Muscular Dystrophy: A Comparative Review.BioDrugs. 2024 Jan;38(1):95-119. doi: 10.1007/s40259-023-00632-3. Epub 2023 Nov 2. BioDrugs. 2024. PMID: 37917377 Free PMC article. Review.
-
Nuclear import by karyopherin-βs: recognition and inhibition.Biochim Biophys Acta. 2011 Sep;1813(9):1593-606. doi: 10.1016/j.bbamcr.2010.10.014. Epub 2010 Oct 26. Biochim Biophys Acta. 2011. PMID: 21029754 Free PMC article. Review.
-
Tenectin is a novel alphaPS2betaPS integrin ligand required for wing morphogenesis and male genital looping in Drosophila.Dev Biol. 2010 Apr 15;340(2):504-17. doi: 10.1016/j.ydbio.2010.02.008. Epub 2010 Feb 10. Dev Biol. 2010. PMID: 20152825 Free PMC article.
-
Regulating Phase Transition in Neurodegenerative Diseases by Nuclear Import Receptors.Biology (Basel). 2022 Jul 4;11(7):1009. doi: 10.3390/biology11071009. Biology (Basel). 2022. PMID: 36101390 Free PMC article. Review.
-
A Tendon Cell Specific RNAi Screen Reveals Novel Candidates Essential for Muscle Tendon Interaction.PLoS One. 2015 Oct 21;10(10):e0140976. doi: 10.1371/journal.pone.0140976. eCollection 2015. PLoS One. 2015. PMID: 26488612 Free PMC article.
References
-
- Allard J. D., Chang H. C., Herbst R., McNeill H., Simon M. A. The SH2-containing tyrosine phosphatase corkscrew is required during signaling by sevenless, Ras1 and Raf. Development. 1996;122:1137–1146. - PubMed
-
- Allard J. D., Herbst R., Carroll P. M., Simon M. A. Mutational analysis of the SRC homology 2 domain protein-tyrosine phosphatase Corkscrew. J. Biol. Chem. 1998;273:13129–13135. - PubMed
-
- Assoian R. K., Schwartz M. A. Coordinate signaling by integrins and receptor tyrosine kinases in the regulation of G1 phase cell-cycle progression. Curr. Opin. Genet. Dev. 2001;11:48–53. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

