Calcineurin in reactive astrocytes plays a key role in the interplay between proinflammatory and anti-inflammatory signals
- PMID: 17699657
- PMCID: PMC6672188
- DOI: 10.1523/JNEUROSCI.1002-07.2007
Calcineurin in reactive astrocytes plays a key role in the interplay between proinflammatory and anti-inflammatory signals
Abstract
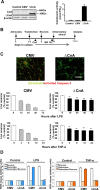
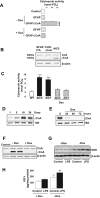
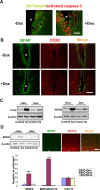
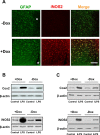
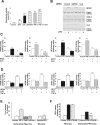
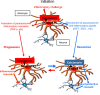
Maladaptive inflammation is a major suspect in progressive neurodegeneration, but the underlying mechanisms are difficult to envisage in part because reactive glial cells at lesion sites secrete both proinflammatory and anti-inflammatory mediators. We now report that astrocytes modulate neuronal resilience to inflammatory insults through the phosphatase calcineurin. In quiescent astrocytes, inflammatory mediators such as tumor necrosis factor-alpha (TNF-alpha) recruits calcineurin to stimulate a canonical inflammatory pathway involving the transcription factors nuclear factor kappaB (NFkappaB) and nuclear factor of activated T-cells (NFAT). However, in reactive astrocytes, local anti-inflammatory mediators such as insulin-like growth factor I also recruit calcineurin but, in this case, to inhibit NFkappaB/NFAT. Proof of concept experiments in vitro showed that expression of constitutively active calcineurin in astrocytes abrogated the inflammatory response after TNF-alpha or endotoxins and markedly enhanced neuronal survival. Furthermore, regulated expression of constitutively active calcineurin in astrocytes markedly reduced inflammatory injury in transgenic mice, in a calcineurin-dependent manner. These results suggest that calcineurin forms part of a molecular pathway whereby reactive astrocytes determine the outcome of the neuroinflammatory process by directing it toward either its resolution or its progression.
Figures

References
-
- Abbasi S, Lee JD, Su B, Chen X, Alcon JL, Yang J, Kellems RE, Xia Y. Protein kinase-mediated regulation of calcineurin through the phosphorylation of modulatory calcineurin-interacting protein 1. J Biol Chem. 2006;281:7717–7726. - PubMed
-
- Alfieri C, Evans-Anderson H, Yutzey K. Developmental regulation of the mouse IGF-I exon 1 promoter region by calcineurin activation of NFAT in skeletal muscle. Am J Physiol Cell Physiol. 2007;292:C1887–C1894. - PubMed
-
- Avni O, Lee D, Macian F, Szabo SJ, Glimcher LH, Rao A. T(H) cell differentiation is accompanied by dynamic changes in histone acetylation of cytokine genes. Nat Immunol. 2002;3:643–651. - PubMed
-
- Bailey ST, Ghosh S. “PPAR”ting ways with inflammation. Nat Immunol. 2005;6:966–967. - PubMed
-
- Beilharz EJ, Russo VC, Butler G, Baker NL, Connor B, Sirimanne ES, Dragunow M, Werther GA, Gluckman PD, Williams CE, Scheepens A. Co-ordinated and cellular specific induction of the components of the IGF/IGFBP axis in the rat brain following hypoxic-ischemic injury. Brain Res Mol Brain Res. 1998;59:119–134. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
