The TRPV3 mutation associated with the hairless phenotype in rodents is constitutively active
- PMID: 17706768
- PMCID: PMC2287378
- DOI: 10.1016/j.ceca.2007.06.004
The TRPV3 mutation associated with the hairless phenotype in rodents is constitutively active
Abstract
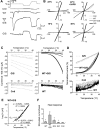
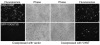
TRPV3 is a non-selective cation channel activated by warm to hot temperatures. In rodents, TRPV3 is highly expressed in basal keratinocytes of skin and oral/nasal epithelia. TRPV3 knockout mice showed impaired responses to innocuous and noxious heat but otherwise normal appearance and reactions to many sensory modalities. However, point mutations of TRPV3 at Gly573 to Ser and Cys have recently been linked to autosomal dominant hairless phenotypes and spontaneous dermatitis in mice and rats, implicating an important role for TRPV3 in alopecia and skin diseases. Exactly, how the mutations affect TRPV3 function was unexplained. Here, we show that both G573S and G573C mutations of murine TRPV3 are constitutively active in heterologous systems. In HEK 293 cells, expression of the TRPV3 mutants causes cell death. In Xenopus oocytes, the constitutively active mutant channel is irresponsive to thermal and chemical stimuli but it reduces the temperature threshold and enhances the responses to heat and TRPV3 agonists of the wild type channel when they are co-expressed. We conclude that the G573S and G573C substitutions render the TRPV3 channel spontaneously active under normal physiological conditions, which in turn alters ion homeostasis and membrane potentials of skin keratinocytes, leading to hair loss and dermatitis-like skin diseases.
Figures




Similar articles
-
Association of a mutation in TRPV3 with defective hair growth in rodents.J Invest Dermatol. 2006 Dec;126(12):2664-72. doi: 10.1038/sj.jid.5700468. Epub 2006 Jul 20. J Invest Dermatol. 2006. PMID: 16858425
-
Voltage- and temperature-dependent activation of TRPV3 channels is potentiated by receptor-mediated PI(4,5)P2 hydrolysis.J Gen Physiol. 2011 Mar;137(3):271-88. doi: 10.1085/jgp.200910388. Epub 2011 Feb 14. J Gen Physiol. 2011. PMID: 21321070 Free PMC article.
-
TRPV3.Handb Exp Pharmacol. 2014;222:273-91. doi: 10.1007/978-3-642-54215-2_11. Handb Exp Pharmacol. 2014. PMID: 24756710 Review.
-
Beyond Ca2+ signalling: the role of TRPV3 in the transport of NH4.Pflugers Arch. 2021 Dec;473(12):1859-1884. doi: 10.1007/s00424-021-02616-0. Epub 2021 Oct 19. Pflugers Arch. 2021. PMID: 34664138 Free PMC article.
-
Novel Insights into the Role of Keratinocytes-Expressed TRPV3 in the Skin.Biomolecules. 2023 Mar 10;13(3):513. doi: 10.3390/biom13030513. Biomolecules. 2023. PMID: 36979447 Free PMC article. Review.
Cited by
-
TRPV3 enhances skin keratinocyte proliferation through EGFR-dependent signaling pathways.Cell Biol Toxicol. 2021 Apr;37(2):313-330. doi: 10.1007/s10565-020-09536-2. Epub 2020 Jun 13. Cell Biol Toxicol. 2021. PMID: 32535744
-
Calcium plays a central role in the sensitization of TRPV3 channel to repetitive stimulations.J Biol Chem. 2008 Mar 7;283(10):6162-74. doi: 10.1074/jbc.M706535200. Epub 2008 Jan 4. J Biol Chem. 2008. PMID: 18178557 Free PMC article.
-
Structural basis for promiscuous action of monoterpenes on TRP channels.Commun Biol. 2021 Mar 5;4(1):293. doi: 10.1038/s42003-021-01776-0. Commun Biol. 2021. PMID: 33674682 Free PMC article.
-
Role of TRPV3 in immune response to development of dermatitis.J Inflamm (Lond). 2009 May 25;6:17. doi: 10.1186/1476-9255-6-17. J Inflamm (Lond). 2009. PMID: 19463197 Free PMC article.
-
Mechanisms of proton inhibition and sensitization of the cation channel TRPV3.J Gen Physiol. 2021 Feb 1;153(2):e202012663. doi: 10.1085/jgp.202012663. J Gen Physiol. 2021. PMID: 33320167 Free PMC article.
References
-
- Montell C, Birnbaumer L, Flockerzi V, et al. A unified nomenclature for the superfamily of TRP cation channels. Mol Cell. 2002;9:229–231. - PubMed
-
- Jordt SE, McKemy DD, Julius D. Lessons from peppers and peppermint: the molecular logic of thermosensation. Curr Opin Neurobiol. 2003;13:487–492. - PubMed
-
- Patapoutian A, Peier AM, Story GM, Viswanath V. ThermoTRP channels and beyond: mechanisms of temperature sensation. Nat Rev Neurosci. 2003;4:529–539. - PubMed
-
- Peier AM, Reeve AJ, Andersson DA, et al. A heat-sensitive TRP channel expressed in keratinocytes. Science. 2002;296:2046–2049. - PubMed
-
- Chung MK, Lee H, Caterina MJ. Warm temperatures activate TRPV4 in mouse 308 keratinocytes. J Biol Chem. 2003;278:32037–32046. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

