Nuclear export of the human cytomegalovirus tegument protein pp65 requires cyclin-dependent kinase activity and the Crm1 exporter
- PMID: 17715235
- PMCID: PMC2168781
- DOI: 10.1128/JVI.02760-06
Nuclear export of the human cytomegalovirus tegument protein pp65 requires cyclin-dependent kinase activity and the Crm1 exporter
Abstract
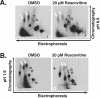
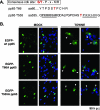
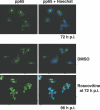
We have previously shown that treatment of human cytomegalovirus-infected cells with the cyclin-dependent kinase (cdk) inhibitor roscovitine has significant effects on several stages of the virus life cycle depending on the time of addition (V. Sanchez, A. K. McElroy, J. Yen, S. Tamrakar, C. L. Clark, R. A. Schwartz, and D. H. Spector, J. Virol. 78:11219-11232, 2004; V. Sanchez and D. Spector, J. Virol. 80:5886-5896, 2006). In this report, we add to these findings by demonstrating alterations in the phosphorylation and localization of pp65 (UL83) in cells treated with roscovitine. We observed that inhibition of cdk activity causes the retention of pp65 within the nucleus at late times postinfection. At the same time, we observed a change in the phosphorylation pattern of the protein. Interestingly, mutation of potential cdk phosphorylation sites did not affect the ability of pp65 to localize to the nucleus or to relocalize to the cytoplasm late in infection. However, we found that the cytoplasmic accumulation of pp65 late in infection was sensitive to the Crm1 inhibitor leptomycin B.
Figures



Similar articles
-
Nucleocytoplasmic shuttling and CRM1-dependent MHC class I peptide presentation of human cytomegalovirus pp65.Med Microbiol Immunol. 2012 Nov;201(4):567-79. doi: 10.1007/s00430-012-0269-7. Epub 2012 Sep 11. Med Microbiol Immunol. 2012. PMID: 22965172
-
Human cytomegalovirus infection induces specific hyperphosphorylation of the carboxyl-terminal domain of the large subunit of RNA polymerase II that is associated with changes in the abundance, activity, and localization of cdk9 and cdk7.J Virol. 2005 Dec;79(24):15477-93. doi: 10.1128/JVI.79.24.15477-15493.2005. J Virol. 2005. PMID: 16306619 Free PMC article.
-
Cyclin A- and cyclin E-Cdk complexes shuttle between the nucleus and the cytoplasm.Mol Biol Cell. 2002 Mar;13(3):1030-45. doi: 10.1091/mbc.01-07-0361. Mol Biol Cell. 2002. PMID: 11907280 Free PMC article.
-
Cyclin/CDK regulates the nucleocytoplasmic localization of the human papillomavirus E1 DNA helicase.J Virol. 2004 Dec;78(24):13954-65. doi: 10.1128/JVI.78.24.13954-13965.2004. J Virol. 2004. PMID: 15564503 Free PMC article.
-
Modification of the major tegument protein pp65 of human cytomegalovirus inhibits virus growth and leads to the enhancement of a protein complex with pUL69 and pUL97 in infected cells.J Gen Virol. 2010 Oct;91(Pt 10):2531-41. doi: 10.1099/vir.0.022293-0. Epub 2010 Jun 30. J Gen Virol. 2010. PMID: 20592110
Cited by
-
Viral Appropriation: Laying Claim to Host Nuclear Transport Machinery.Cells. 2019 Jun 8;8(6):559. doi: 10.3390/cells8060559. Cells. 2019. PMID: 31181773 Free PMC article. Review.
-
The absence of p53 during Human Cytomegalovirus infection leads to decreased UL53 expression, disrupting UL50 localization to the inner nuclear membrane, and thereby inhibiting capsid nuclear egress.Virology. 2016 Oct;497:262-278. doi: 10.1016/j.virol.2016.07.020. Epub 2016 Aug 4. Virology. 2016. PMID: 27498409 Free PMC article.
-
Human cytomegaloviruses expressing yellow fluorescent fusion proteins--characterization and use in antiviral screening.PLoS One. 2010 Feb 11;5(2):e9174. doi: 10.1371/journal.pone.0009174. PLoS One. 2010. PMID: 20161802 Free PMC article.
-
The Second-Generation XPO1 Inhibitor Eltanexor Inhibits Human Cytomegalovirus (HCMV) Replication and Promotes Type I Interferon Response.Front Microbiol. 2021 May 3;12:675112. doi: 10.3389/fmicb.2021.675112. eCollection 2021. Front Microbiol. 2021. PMID: 34012430 Free PMC article.
-
Biologic and immunologic effects of knockout of human cytomegalovirus pp65 nuclear localization signal.Clin Vaccine Immunol. 2009 Jun;16(6):935-43. doi: 10.1128/CVI.00011-09. Epub 2009 Apr 15. Clin Vaccine Immunol. 2009. PMID: 19369477 Free PMC article.
References
-
- Blom, N., S. Gammeltoft, and S. Brunak. 1999. Sequence and structure-based prediction of eukaryotic protein phosphorylation sites. J. Mol. Biol. 294:1351-1362. - PubMed
-
- Bresnahan, W. A., I. Boldogh, P. Chi, E. A. Thompson, and T. Albrecht. 1997. Inhibition of cellular Cdk2 activity blocks human cytomegalovirus replication. Virology 231:239-247. - PubMed
-
- Cinatl, J., Jr., J.-U. Vogel, R. Kotchetknov, and H. W. Doerr. 2004. Oncomodulatory signals by regulatory proteins encoded by human cytomegalovirus: a novel role for viral infection in tumor progression. FEMS Microbiol. Rev. 28:59-77. - PubMed
-
- Dal Monte, P., S. Pignatelli, N. Zini, N. Maraldi, E. Perret, M. Prevost, and M. Landini. 2002. Analysis of intracellular and intraviral localization of the human cytomegalovirus UL53 protein. J. Gen. Virol. 83:1005-1012. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

