Inhaled NO accelerates restoration of liver function in adults following orthotopic liver transplantation
- PMID: 17717604
- PMCID: PMC1950460
- DOI: 10.1172/JCI31892
Inhaled NO accelerates restoration of liver function in adults following orthotopic liver transplantation
Abstract
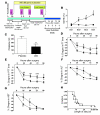
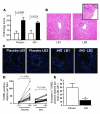
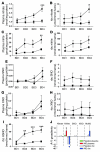
Ischemia/reperfusion (IR) injury in transplanted livers contributes to organ dysfunction and failure and is characterized in part by loss of NO bioavailability. Inhalation of NO is nontoxic and at high concentrations (80 ppm) inhibits IR injury in extrapulmonary tissues. In this prospective, blinded, placebo-controlled study, we evaluated the hypothesis that administration of inhaled NO (iNO; 80 ppm) to patients undergoing orthotopic liver transplantation inhibits hepatic IR injury, resulting in improved liver function. Patients were randomized to receive either placebo or iNO (n = 10 per group) during the operative period only. When results were adjusted for cold ischemia time and sex, iNO significantly decreased hospital length of stay, and evaluation of serum transaminases (alanine transaminase, aspartate aminotransferase) and coagulation times (prothrombin time, partial thromboplastin time) indicated that iNO improved the rate at which liver function was restored after transplantation. iNO did not significantly affect changes in inflammatory markers in liver tissue 1 hour after reperfusion but significantly lowered hepatocyte apoptosis. Evaluation of circulating NO metabolites indicated that the most likely candidate transducer of extrapulmonary effects of iNO was nitrite. In summary, this study supports the clinical use of iNO as an extrapulmonary therapeutic to improve organ function following transplantation.
Figures

Comment in
-
A simple treatment to prevent ischemia-reperfusion injury of the liver.Liver Transpl. 2007 Dec;13(12):1755. Liver Transpl. 2007. PMID: 18178986 No abstract available.
-
The inhaled route to improved liver function.Hepatology. 2008 Feb;47(2):755-6. doi: 10.1002/hep.22190. Hepatology. 2008. PMID: 18220294 No abstract available.
Similar articles
-
Inhaled nitric oxide after left ventricular assist device implantation: a prospective, randomized, double-blind, multicenter, placebo-controlled trial.J Heart Lung Transplant. 2011 Aug;30(8):870-8. doi: 10.1016/j.healun.2011.03.005. Epub 2011 Apr 29. J Heart Lung Transplant. 2011. PMID: 21530317 Clinical Trial.
-
Beneficial effects of inhaled nitric oxide with intravenous steroid in an ischemia-reperfusion model involving aortic clamping.Int J Immunopathol Pharmacol. 2018 Jan-Dec;32:394632017751486. doi: 10.1177/0394632017751486. Int J Immunopathol Pharmacol. 2018. PMID: 29376749 Free PMC article.
-
A randomized clinical trial testing the anti-inflammatory effects of preemptive inhaled nitric oxide in human liver transplantation.PLoS One. 2014 Feb 12;9(2):e86053. doi: 10.1371/journal.pone.0086053. eCollection 2014. PLoS One. 2014. PMID: 24533048 Free PMC article. Clinical Trial.
-
Role of inhaled nitric oxide in adult heart or lung transplant recipients.Ann Pharmacother. 2005 May;39(5):913-7. doi: 10.1345/aph.1E228. Epub 2005 Mar 15. Ann Pharmacother. 2005. PMID: 15769831 Review.
-
The effects of inhaled nitric oxide after lung transplantation.Minerva Anestesiol. 2010 May;76(5):353-61. Minerva Anestesiol. 2010. PMID: 20395898 Review.
Cited by
-
Changing Perspectives from Oxidative Stress to Redox Signaling-Extracellular Redox Control in Translational Medicine.Antioxidants (Basel). 2022 Jun 16;11(6):1181. doi: 10.3390/antiox11061181. Antioxidants (Basel). 2022. PMID: 35740078 Free PMC article. Review.
-
Nitric oxide signalling and neuronal nitric oxide synthase in the heart under stress.F1000Res. 2017 May 23;6:742. doi: 10.12688/f1000research.10128.1. eCollection 2017. F1000Res. 2017. PMID: 28649367 Free PMC article. Review.
-
Mitochondrial dysfunction and oxidative stress in the pathogenesis of alcohol- and obesity-induced fatty liver diseases.Free Radic Biol Med. 2008 Apr 1;44(7):1259-72. doi: 10.1016/j.freeradbiomed.2007.12.029. Epub 2008 Jan 3. Free Radic Biol Med. 2008. PMID: 18242193 Free PMC article. Review.
-
Improving outcomes after cardiac arrest using NO inhalation.Trends Cardiovasc Med. 2013 Feb;23(2):52-8. doi: 10.1016/j.tcm.2012.08.011. Epub 2013 Jan 3. Trends Cardiovasc Med. 2013. PMID: 23291033 Free PMC article. Review.
-
Nitrite in organ protection.Br J Pharmacol. 2014 Jan;171(1):1-11. doi: 10.1111/bph.12291. Br J Pharmacol. 2014. PMID: 23826831 Free PMC article. Review.
References
-
- Urakami H., Grisham M.B. Divergent roles of superoxide and nitric oxide in reduced-size liver ischemia and reperfusion injury: implications for partial liver transplantation. Pathophysiology. 2006;13:183–193. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

