Expression of a CD20-specific chimeric antigen receptor enhances cytotoxic activity of NK cells and overcomes NK-resistance of lymphoma and leukemia cells
- PMID: 17717662
- PMCID: PMC11029838
- DOI: 10.1007/s00262-007-0383-3
Expression of a CD20-specific chimeric antigen receptor enhances cytotoxic activity of NK cells and overcomes NK-resistance of lymphoma and leukemia cells
Abstract
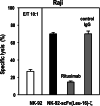
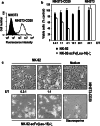
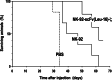
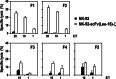
Despite the clinical success of CD20-specific antibody rituximab, malignancies of B-cell origin continue to present a major clinical challenge, in part due to an inability of the antibody to activate antibody-dependent cell-mediated cytotoxicity (ADCC) in some patients, and development of resistance in others. Expression of chimeric antigen receptors in effector cells operative in ADCC might allow to bypass insufficient activation via FcgammaRIII and other resistance mechanisms that limit natural killer (NK)-cell activity. Here we have generated genetically modified NK cells carrying a chimeric antigen receptor that consists of a CD20-specific scFv antibody fragment, via a flexible hinge region connected to the CD3zeta chain as a signaling moiety. As effector cells we employed continuously growing, clinically applicable human NK-92 cells. While activity of the retargeted NK-92 against CD20-negative targets remained unchanged, the gene modified NK cells displayed markedly enhanced cytotoxicity toward NK-sensitive CD20 expressing cells. Importantly, in contrast to parental NK-92, CD20-specific NK cells efficiently lysed CD20 expressing but otherwise NK-resistant established and primary lymphoma and leukemia cells, demonstrating that this strategy can overcome NK-cell resistance and might be suitable for the development of effective cell-based therapeutics for the treatment of B-cell malignancies.
Figures



References
-
- Maloney DG, Liles TM, Czerwinski DK, Waldichuk C, Rosenberg J, Grillo-Lopez A, Levy R. Phase I clinical trial using escalating single-dose infusion of chimeric anti-CD20 monoclonal antibody (IDEC-C2B8) in patients with recurrent B-cell lymphoma. Blood. 1994;84:2457–2466. - PubMed
-
- Maloney DG, Grillo-Lopez AJ, White CA, Bodkin D, Schilder RJ, Neidhart JA, Janakiraman N, Foon KA, Liles TM, Dallaire BK, Wey K, Royston I, Davis T, Levy R. IDEC-C2B8 (Rituximab) anti-CD20 monoclonal antibody therapy in patients with relapsed low-grade non-Hodgkin’s lymphoma. Blood. 1997;90:2188–2195. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

