Repeated amphetamine administration outside the home cage enhances drug-induced Fos expression in rat nucleus accumbens
- PMID: 17720257
- PMCID: PMC2135552
- DOI: 10.1016/j.bbr.2007.07.024
Repeated amphetamine administration outside the home cage enhances drug-induced Fos expression in rat nucleus accumbens
Abstract
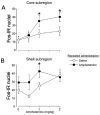
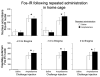
Induction of the immediate early gene protein product Fos has been used extensively to assess neural activation in the striatum after repeated amphetamine administration to rats in their home cages. However, this technique has not been used to examine striatal activation after repeated administration outside the home cage, an environment where repeated drug administration produces more robust psychomotor sensitization. We determined the dose-response relationship for amphetamine-induced psychomotor activity and Fos expression in nucleus accumbens and caudate-putamen 1 week after repeated administration of amphetamine or saline in locomotor activity chambers. Repeated administration of amphetamine enhanced amphetamine-induced locomotor activity and stereotypy and Fos expression in nucleus accumbens, but not in caudate-putamen. In comparison, levels of Fos expression induced by 1mg/kg amphetamine were not altered in nucleus accumbens or caudate-putamen by repeated amphetamine administration in the home cage. Double-labeling of Fos protein and enkephalin mRNA indicates that Fos is expressed in approximately equal numbers of enkephalin-negative and enkephalin-positive neurons in nucleus accumbens and caudate-putamen following injections outside the home cage. Furthermore, repeated amphetamine administration increased drug-induced Fos expression in enkephalin-positive, but not enkephalin-negative, neurons in nucleus accumbens. We conclude that repeated amphetamine administration outside the home cage recruits the activation of enkephalin-containing nucleus accumbens neurons during sensitized amphetamine-induced psychomotor activity.
Figures





Similar articles
-
Cocaine-induced locomotor activity and Fos expression in nucleus accumbens are sensitized for 6 months after repeated cocaine administration outside the home cage.Eur J Neurosci. 2006 Aug;24(3):867-75. doi: 10.1111/j.1460-9568.2006.04969.x. Eur J Neurosci. 2006. PMID: 16930414
-
Expression of amphetamine sensitization is associated with recruitment of a reactive neuronal population in the nucleus accumbens core.Psychopharmacology (Berl). 2008 May;198(1):113-26. doi: 10.1007/s00213-008-1100-4. Epub 2008 Mar 18. Psychopharmacology (Berl). 2008. PMID: 18347780 Free PMC article.
-
Context-specific sensitization of cocaine-induced locomotor activity and associated neuronal ensembles in rat nucleus accumbens.Eur J Neurosci. 2008 Jan;27(1):202-12. doi: 10.1111/j.1460-9568.2007.05984.x. Epub 2007 Dec 17. Eur J Neurosci. 2008. PMID: 18093170
-
Compartment-specific changes in striatal neuronal activity during expression of amphetamine sensitization are the result of drug hypersensitivity.Eur J Neurosci. 2002 Dec;16(12):2462-8. doi: 10.1046/j.1460-9568.2002.02308.x. Eur J Neurosci. 2002. PMID: 12492441
-
Differences in the regional and cellular localization of c-fos messenger RNA induced by amphetamine, cocaine and caffeine in the rat.Neuroscience. 1994 Apr;59(4):837-49. doi: 10.1016/0306-4522(94)90288-7. Neuroscience. 1994. PMID: 7520134
Cited by
-
Prenatal Amphetamine-Induced Dopaminergic Alteration in a Gender- and Estrogen-Dependent Manner.Neurochem Res. 2022 May;47(5):1317-1328. doi: 10.1007/s11064-022-03531-1. Epub 2022 Feb 11. Neurochem Res. 2022. PMID: 35147850
-
Regional Differences in Striatal Neuronal Ensemble Excitability Following Cocaine and Extinction Memory Retrieval in Fos-GFP Mice.Neuropsychopharmacology. 2018 Mar;43(4):718-727. doi: 10.1038/npp.2017.101. Epub 2017 May 25. Neuropsychopharmacology. 2018. PMID: 28540927 Free PMC article.
-
Selective bilateral lesion to caudate nucleus modulates the acute and chronic methylphenidate effects.Pharmacol Biochem Behav. 2012 Apr;101(2):208-16. doi: 10.1016/j.pbb.2012.01.002. Epub 2012 Jan 11. Pharmacol Biochem Behav. 2012. PMID: 22260871 Free PMC article.
-
Persistence of one-trial cocaine-induced behavioral sensitization in young rats: regional differences in Fos immunoreactivity.Psychopharmacology (Berl). 2009 Apr;203(3):617-28. doi: 10.1007/s00213-008-1407-1. Epub 2008 Nov 20. Psychopharmacology (Berl). 2009. PMID: 19020866
-
Identification and Verification of Potential Hub Genes in Amphetamine-Type Stimulant (ATS) and Opioid Dependence by Bioinformatic Analysis.Front Genet. 2022 Mar 30;13:837123. doi: 10.3389/fgene.2022.837123. eCollection 2022. Front Genet. 2022. PMID: 35432486 Free PMC article.
References
-
- Angulo JA. Involvement of dopamine D1 and D2 receptors in the regulation of proenkephalin mRNA abundance in the striatum and accumbens of the rat brain. J Neurochem. 1992;58:1104–9. - PubMed
-
- Backman C, et al. Cellular mRNA expression of the transcription factor NGFI-B suggests a gene regulatory role in striatal opiate-peptide neurons. Brain Res. 2001;903:26–32. - PubMed
-
- Badiani A, et al. The development of sensitization to the psychomotor stimulant effects of amphetamine is enhanced in a novel environment. Psychopharmacology (Berl) 1995a;117:443–52. - PubMed
-
- Badiani A, et al. Influence of novel versus home environments on sensitization to the psychomotor stimulant effects of cocaine and amphetamine. Brain Res. 1995b;674:291–8. - PubMed
-
- Badiani A, et al. Enduring enhancement of amphetamine sensitization by drug-associated environmental stimuli. J Pharmacol Exp Ther. 1997;282:787–94. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

