Localization and visualization of a coxiella-type symbiont within the lone star tick, Amblyomma americanum
- PMID: 17720830
- PMCID: PMC2075054
- DOI: 10.1128/AEM.00537-07
Localization and visualization of a coxiella-type symbiont within the lone star tick, Amblyomma americanum
Abstract
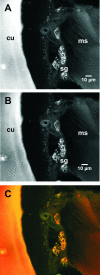
A Coxiella-type microbe occurs at 100% frequency in all Amblyomma americanum ticks thus far tested. Using laboratory-reared ticks free of other microbes, we identified the Amblyomma-associated Coxiella microbe in several types of tissue and at various stages of the life cycle of A. americanum by 16S rRNA gene sequencing and diagnostic PCR. We visualized Amblyomma-associated Coxiella through the use of a diagnostic fluorescence in situ hybridization (FISH) assay supplemented with PCR-based detection, nucleic acid fluorescent staining, wide-field epifluorescence and confocal microscopy, and transmission electron microscopy (TEM). Specific fluorescent foci were observed in several tick tissues, including the midgut and the Malpighian tubules, but particularly bright signals were observed in the granular acini of salivary gland clusters and in both small and large oocytes. TEM confirmed intracellular bacterial structures in the same tissues. The presence of Amblyomma-associated Coxiella within oocytes is consistent with the vertical transmission of these endosymbionts. Further, the presence of the Amblyomma-associated Coxiella symbiont in other tissues such as salivary glands could potentially lead to interactions with horizontally acquired pathogens.
Figures




Similar articles
-
Tissue localization of Coxiella-like endosymbionts in three European tick species through fluorescence in situ hybridization.Ticks Tick Borne Dis. 2019 Jun;10(4):798-804. doi: 10.1016/j.ttbdis.2019.03.014. Epub 2019 Mar 19. Ticks Tick Borne Dis. 2019. PMID: 30922601
-
A Coxiella-like endosymbiont is a potential vitamin source for the Lone Star tick.Genome Biol Evol. 2015 Jan 23;7(3):831-8. doi: 10.1093/gbe/evv016. Genome Biol Evol. 2015. PMID: 25618142 Free PMC article.
-
Highly prevalent Coxiella sp. bacterium in the tick vector Amblyomma americanum.Appl Environ Microbiol. 2007 Jan;73(1):334-6. doi: 10.1128/AEM.02009-06. Epub 2006 Nov 3. Appl Environ Microbiol. 2007. PMID: 17085709 Free PMC article.
-
Coxiella-like endosymbionts.Adv Exp Med Biol. 2012;984:365-79. doi: 10.1007/978-94-007-4315-1_18. Adv Exp Med Biol. 2012. PMID: 22711641 Review.
-
Role of the lone star tick, Amblyomma americanum (L.), in human and animal diseases.Vet Parasitol. 2009 Mar 9;160(1-2):1-12. doi: 10.1016/j.vetpar.2008.10.089. Epub 2008 Oct 28. Vet Parasitol. 2009. PMID: 19054615 Review.
Cited by
-
The Symbiotic Continuum Within Ticks: Opportunities for Disease Control.Front Microbiol. 2022 Mar 17;13:854803. doi: 10.3389/fmicb.2022.854803. eCollection 2022. Front Microbiol. 2022. PMID: 35369485 Free PMC article. Review.
-
Comparative genomics reveal extensive transposon-mediated genomic plasticity and diversity among potential effector proteins within the genus Coxiella.Infect Immun. 2009 Feb;77(2):642-56. doi: 10.1128/IAI.01141-08. Epub 2008 Dec 1. Infect Immun. 2009. PMID: 19047403 Free PMC article.
-
The bacterial community of the lone star tick (Amblyomma americanum).Parasit Vectors. 2021 Jan 14;14(1):49. doi: 10.1186/s13071-020-04550-z. Parasit Vectors. 2021. PMID: 33446262 Free PMC article.
-
Coxiellaceae in Ticks from Human, Domestic and Wild Hosts from Sardinia, Italy: High Diversity of Coxiella-like Endosymbionts.Acta Parasitol. 2021 Jun;66(2):654-663. doi: 10.1007/s11686-020-00324-w. Epub 2021 Jan 25. Acta Parasitol. 2021. PMID: 33492605
-
Coxiella burnetii infections from animals and ticks in South Africa: a systematic review.Vet Res Commun. 2024 Feb;48(1):19-28. doi: 10.1007/s11259-023-10204-z. Epub 2023 Aug 29. Vet Res Commun. 2024. PMID: 37642820 Free PMC article.
References
-
- Babudieri, B. 1959. Q fever: a zoonosis. Adv. Vet. Sci. 5:81-182.
-
- Balashov, Y. S. 1972. Bloodsucking ticks (Ixodoidea): vectors of disease of man and animals. Misc. Publ. Entomol. Soc. Amer. 8:161-376.
-
- Barrett, J. M., P. M. Heidger, and S. W. Kennedy. 1975. Chelated bismuth as a stain in electron microscopy. J. Histochem. Cytochem. 23:780-783. - PubMed
-
- Bernasconi, M. V., S. Casati, O. Peter, and J. C. Piffaretti. 2002. Rhipicephalus ticks infected with Rickettsia and Coxiella in Southern Switzerland (Canton Ticino). Infect. Genet. Evol. 2:111-120. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

