Nanofibers and their applications in tissue engineering
- PMID: 17722259
- PMCID: PMC2426767
- DOI: 10.2147/nano.2006.1.1.15
Nanofibers and their applications in tissue engineering
Abstract
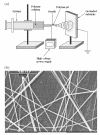
Developing scaffolds that mimic the architecture of tissue at the nanoscale is one of the major challenges in the field of tissue engineering. The development of nanofibers has greatly enhanced the scope for fabricating scaffolds that can potentially meet this challenge. Currently, there are three techniques available for the synthesis of nanofibers: electrospinning, self-assembly, and phase separation. Of these techniques, electrospinning is the most widely studied technique and has also demonstrated the most promising results in terms of tissue engineering applications. The availability of a wide range of natural and synthetic biomaterials has broadened the scope for development of nanofibrous scaffolds, especially using the electrospinning technique. The three dimensional synthetic biodegradable scaffolds designed using nanofibers serve as an excellent framework for cell adhesion, proliferation, and differentiation. Therefore, nanofibers, irrespective of their method of synthesis, have been used as scaffolds for musculoskeletal tissue engineering (including bone, cartilage, ligament, and skeletal muscle), skin tissue engineering, vascular tissue engineering, neural tissue engineering, and as carriers for the controlled delivery of drugs, proteins, and DNA. This review summarizes the currently available techniques for nanofiber synthesis and discusses the use of nanofibers in tissue engineering and drug delivery applications.
Figures



Similar articles
-
Therapeutic application of electrospun nanofibrous meshes.Nanomedicine (Lond). 2014 Apr;9(4):517-33. doi: 10.2217/nnm.13.224. Nanomedicine (Lond). 2014. PMID: 24787440 Review.
-
Recent development of polymer nanofibers for biomedical and biotechnological applications.J Mater Sci Mater Med. 2005 Oct;16(10):933-46. doi: 10.1007/s10856-005-4428-x. J Mater Sci Mater Med. 2005. PMID: 16167102 Review.
-
Recent advances in polymer nanofibers.J Nanosci Nanotechnol. 2004 Jan-Feb;4(1-2):52-65. J Nanosci Nanotechnol. 2004. PMID: 15112541 Review.
-
Electrospun nanofiber fabrication as synthetic extracellular matrix and its potential for vascular tissue engineering.Tissue Eng. 2004 Jul-Aug;10(7-8):1160-8. doi: 10.1089/ten.2004.10.1160. Tissue Eng. 2004. PMID: 15363172
-
Biodegradable nanomats produced by electrospinning: expanding multifunctionality and potential for tissue engineering.J Nanosci Nanotechnol. 2006 Sep-Oct;6(9-10):2693-711. doi: 10.1166/jnn.2006.485. J Nanosci Nanotechnol. 2006. PMID: 17048476 Review.
Cited by
-
Nanotechnology-Based Drug Delivery Systems for Melanoma Antitumoral Therapy: A Review.Biomed Res Int. 2015;2015:841817. doi: 10.1155/2015/841817. Epub 2015 May 11. Biomed Res Int. 2015. PMID: 26078967 Free PMC article. Review.
-
Biomedical Applications of Magnetically Functionalized Organic/Inorganic Hybrid Nanofibers.Int J Mol Sci. 2015 Jun 15;16(6):13661-77. doi: 10.3390/ijms160613661. Int J Mol Sci. 2015. PMID: 26084046 Free PMC article. Review.
-
Nanofibers prepared by needleless electrospinning technology as scaffolds for wound healing.J Mater Sci Mater Med. 2012 Apr;23(4):931-41. doi: 10.1007/s10856-012-4577-7. Epub 2012 Feb 14. J Mater Sci Mater Med. 2012. PMID: 22331377
-
A Review of Chitosan and Chitosan Nanofiber: Preparation, Characterization, and Its Potential Applications.Polymers (Basel). 2023 Jun 26;15(13):2820. doi: 10.3390/polym15132820. Polymers (Basel). 2023. PMID: 37447465 Free PMC article. Review.
-
Nanoparticles as Drug Delivery Systems in Cancer Medicine: Emphasis on RNAi-Containing Nanoliposomes.Pharmaceuticals (Basel). 2013 Nov 4;6(11):1361-80. doi: 10.3390/ph6111361. Pharmaceuticals (Basel). 2013. PMID: 24287462 Free PMC article.
References
-
- Atala A, Lanza RP. Methods of tissue engineering. San Diego: Academic Pr; 2002.
-
- Berndt P, Fields GB, Tirrell M. Synthetic lipidation of peptides and amino acids: monolayer structure and properties. J Am Chem Soc. 1995;117:9515–22.
-
- Bhattarai SR, Bhattarai N, Yi HK, et al. Novel biodegradable electrospun membrane: scaffold for tissue engineering. Biomaterials. 2004;25:2595–602. - PubMed
-
- Bhattarai N, Edmondson D, Veiseh O, et al. Electrospun chitosan-based nanofibers and their cellular compatibility. Biomaterials. 2005 In press. - PubMed
-
- Boland ED, Matthews JA, Pawlowski KJ, et al. Electrospinning collagen and elastin: preliminary vascular tissue engineering. Front Biosci. 2004;9:1422–32. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

