Central serotonergic neurons are differentially required for opioid analgesia but not for morphine tolerance or morphine reward
- PMID: 17724336
- PMCID: PMC1964813
- DOI: 10.1073/pnas.0705740104
Central serotonergic neurons are differentially required for opioid analgesia but not for morphine tolerance or morphine reward
Abstract
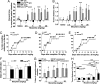
Opioids remain the most effective analgesics despite their potential adverse effects such as tolerance and addiction. Mechanisms underlying these opiate-mediated processes remain the subject of much debate. Here we describe opioid-induced behaviors of Lmx1b conditional knockout mice (Lmx1bf/f/p), which lack central serotonergic neurons, and we report that opioid analgesia is differentially dependent on the central serotonergic system. Analgesia induced by a kappa opioid receptor agonist administered at the supraspinal level was abolished in Lmx1bf/f/p mice compared with their wild-type littermates. Furthermore, compared with their wild-type littermates Lmx1bf/f/p mice exhibited significantly reduced analgesic effects of mu and delta opioid receptor agonists at both spinal and supraspinal sites. In contrast to the attenuation in opioid analgesia, Lmx1bf/f/p mice developed tolerance to morphine analgesia and displayed normal morphine reward behavior as measured by conditioned place preference. Our results provide genetic evidence supporting the view that the central serotonergic system is a key component of supraspinal pain modulatory circuitry mediating opioid analgesia. Furthermore, our data suggest that the mechanisms of morphine tolerance and morphine reward are independent of the central serotonergic system.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

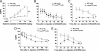


Similar articles
-
Morphine can produce analgesia via spinal kappa opioid receptors in the absence of mu opioid receptors.Brain Res. 2006 Apr 14;1083(1):61-9. doi: 10.1016/j.brainres.2006.01.095. Epub 2006 Mar 10. Brain Res. 2006. PMID: 16530171
-
T394A Mutation at the μ Opioid Receptor Blocks Opioid Tolerance and Increases Vulnerability to Heroin Self-Administration in Mice.J Neurosci. 2016 Oct 5;36(40):10392-10403. doi: 10.1523/JNEUROSCI.0603-16.2016. J Neurosci. 2016. PMID: 27707973 Free PMC article.
-
Loss of morphine reward and dependence in mice lacking G protein-coupled receptor kinase 5.Biol Psychiatry. 2014 Nov 15;76(10):767-74. doi: 10.1016/j.biopsych.2014.01.021. Epub 2014 Feb 3. Biol Psychiatry. 2014. PMID: 24629717 Free PMC article.
-
Role of delivery and trafficking of delta-opioid peptide receptors in opioid analgesia and tolerance.Trends Pharmacol Sci. 2006 Jun;27(6):324-9. doi: 10.1016/j.tips.2006.04.005. Epub 2006 May 6. Trends Pharmacol Sci. 2006. PMID: 16678916 Review.
-
Possible mechanisms of morphine analgesia.Clin Neuropharmacol. 1991 Apr;14(2):131-47. doi: 10.1097/00002826-199104000-00003. Clin Neuropharmacol. 1991. PMID: 1849794 Review.
Cited by
-
Preclinical and early clinical investigations related to monoaminergic pain modulation.Neurotherapeutics. 2009 Oct;6(4):703-12. doi: 10.1016/j.nurt.2009.07.009. Neurotherapeutics. 2009. PMID: 19789074 Free PMC article. Review.
-
Utility of genetically modified mice for understanding the neurobiology of substance use disorders.Hum Genet. 2012 Jun;131(6):941-57. doi: 10.1007/s00439-011-1129-z. Epub 2011 Dec 22. Hum Genet. 2012. PMID: 22190154 Free PMC article. Review.
-
Tetracycline inducible gene manipulation in serotonergic neurons.PLoS One. 2012;7(5):e38193. doi: 10.1371/journal.pone.0038193. Epub 2012 May 31. PLoS One. 2012. PMID: 22693598 Free PMC article.
-
Lmx1b is required at multiple stages to build expansive serotonergic axon architectures.Elife. 2019 Jul 29;8:e48788. doi: 10.7554/eLife.48788. Elife. 2019. PMID: 31355748 Free PMC article.
-
Selective p38α MAPK deletion in serotonergic neurons produces stress resilience in models of depression and addiction.Neuron. 2011 Aug 11;71(3):498-511. doi: 10.1016/j.neuron.2011.06.011. Neuron. 2011. PMID: 21835346 Free PMC article.
References
-
- Millan MJ. Prog Neurobiol. 2002;66:355–474. - PubMed
-
- Le Bars D. In: Neuronal Serotonin. Osborne NN, Hamon M, editors. New York: Wiley; 1988. pp. 171–229.
-
- Sawynok J. Can J Physiol Pharmacol. 1989;67:975–988. - PubMed
-
- Hammond DL. In: Serotonin and Pain. Besson JM, editor. Amsterdam: Elsevier Science; 1990. pp. 251–261.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

