Synergism of Bacillus thuringiensis toxins by a fragment of a toxin-binding cadherin
- PMID: 17724346
- PMCID: PMC1955780
- DOI: 10.1073/pnas.0706011104
Synergism of Bacillus thuringiensis toxins by a fragment of a toxin-binding cadherin
Abstract
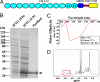
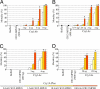
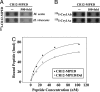
The insecticidal crystal proteins produced by Bacillus thuringiensis (Bt) are broadly used to control insect pests with agricultural importance. The cadherin Bt-R(1) is a binding protein for Bt Cry1A toxins in midgut epithelia of tobacco hornworm (Manduca sexta). We previously identified the Bt-R(1) region most proximal to the cell membrane (CR12-MPED) as the essential binding region required for Cry1Ab-mediated cytotoxicity. Here, we report that a peptide containing this region expressed in Escherichia coli functions as a synergist of Cry1A toxicity against lepidopteran larvae. Far-UV circular dichroism and (1)H-NMR spectroscopy confirmed that our purified CR12-MPED peptide mainly consisted of beta-strands and random coils with unfolded structure. CR12-MPED peptide bound brush border membrane vesicles with high affinity (K(d) = 32 nM) and insect midgut microvilli but did not alter Cry1Ab or Cry1Ac binding localization in the midgut. By BIAcore analysis we demonstrate that Cry1Ab binds CR12-MPED at high (9 nM)- and low (1 microM)-affinity sites. CR12-MPED-mediated Cry1A toxicity enhancement was significantly reduced when the high-affinity Cry1A-binding epitope ((1416)GVLTLNIQ(1423)) within the peptide was altered. Because the mixtures of low Bt toxin dose and CR12-MPED peptide effectively control target insect pests, our discovery has important implications related to the use of this peptide to enhance insecticidal activity of Bt toxin-based biopesticides and transgenic Bt crops.
Conflict of interest statement
Conflict of interest statement: J.C., G.H., M.A.A., and M.J.A. are coinventors on the U.S. patent application Peptides for Inhibiting Insects (US 20050283857). M.J.A. is a founder of InsectiGen, a start-up biotech company in Athens, GA, that is developing and commercializing this technology and is chief scientific officer of InsectiGen. M.A.A. is currently employed by InsectiGen.
Figures

Similar articles
-
Helicoverpa armigera cadherin fragment enhances Cry1Ac insecticidal activity by facilitating toxin-oligomer formation.Appl Microbiol Biotechnol. 2010 Jan;85(4):1033-40. doi: 10.1007/s00253-009-2142-1. Epub 2009 Aug 4. Appl Microbiol Biotechnol. 2010. PMID: 19652967
-
Mapping the epitope in cadherin-like receptors involved in Bacillus thuringiensis Cry1A toxin interaction using phage display.J Biol Chem. 2001 Aug 3;276(31):28906-12. doi: 10.1074/jbc.M103007200. Epub 2001 May 30. J Biol Chem. 2001. PMID: 11384982
-
Enhancement of insecticidal activity of Bacillus thuringiensis Cry1A toxins by fragments of a toxin-binding cadherin correlates with oligomer formation.Peptides. 2009 Mar;30(3):583-8. doi: 10.1016/j.peptides.2008.08.006. Epub 2008 Aug 20. Peptides. 2009. PMID: 18778745 Free PMC article.
-
The pre-pore from Bacillus thuringiensis Cry1Ab toxin is necessary to induce insect death in Manduca sexta.Peptides. 2008 Feb;29(2):318-23. doi: 10.1016/j.peptides.2007.09.026. Epub 2007 Dec 14. Peptides. 2008. PMID: 18226424 Free PMC article. Review.
-
Role of receptor interaction in the mode of action of insecticidal Cry and Cyt toxins produced by Bacillus thuringiensis.Peptides. 2007 Jan;28(1):169-73. doi: 10.1016/j.peptides.2006.06.013. Epub 2006 Dec 4. Peptides. 2007. PMID: 17145116 Review.
Cited by
-
Domain II loop 3 of Bacillus thuringiensis Cry1Ab toxin is involved in a "ping pong" binding mechanism with Manduca sexta aminopeptidase-N and cadherin receptors.J Biol Chem. 2009 Nov 20;284(47):32750-7. doi: 10.1074/jbc.M109.024968. Epub 2009 Oct 6. J Biol Chem. 2009. PMID: 19808680 Free PMC article.
-
Differential protection of Cry1Fa toxin against Spodoptera frugiperda larval gut proteases by cadherin orthologs correlates with increased synergism.Appl Environ Microbiol. 2012 Jan;78(2):354-62. doi: 10.1128/AEM.06212-11. Epub 2011 Nov 11. Appl Environ Microbiol. 2012. PMID: 22081566 Free PMC article.
-
Anopheles gambiae cadherin AgCad1 binds the Cry4Ba toxin of Bacillus thuringiensis israelensis and a fragment of AgCad1 synergizes toxicity.Biochemistry. 2008 May 6;47(18):5101-10. doi: 10.1021/bi7023578. Epub 2008 Apr 12. Biochemistry. 2008. PMID: 18407662 Free PMC article.
-
Aedes aegypti cadherin serves as a putative receptor of the Cry11Aa toxin from Bacillus thuringiensis subsp. israelensis.Biochem J. 2009 Nov 11;424(2):191-200. doi: 10.1042/BJ20090730. Biochem J. 2009. PMID: 19732034 Free PMC article.
-
Enhancement of Bacillus thuringiensis Cry3Aa and Cry3Bb toxicities to coleopteran larvae by a toxin-binding fragment of an insect cadherin.Appl Environ Microbiol. 2009 May;75(10):3086-92. doi: 10.1128/AEM.00268-09. Epub 2009 Mar 27. Appl Environ Microbiol. 2009. PMID: 19329664 Free PMC article.
References
-
- Brickle DS, Turnipseed SG, Sullivan MJ. J Econ Entomol. 2001;94:86–92. - PubMed
-
- Bravo A, Gomez I, Conde J, Munoz-Garay C, Sanchez J, Miranda R, Zhuang M, Gill SS, Soberon M. Biochim Biophys Acta. 2004;1667:38–46. - PubMed
-
- Zhang X, Candas M, Griko NB, Rose-Young L, Bulla LA., Jr Cell Death Differ. 2005;12:1407–1416. - PubMed
-
- Dorsch JA, Candas M, Griko NB, Maaty WSA, Midboe EG, Vadlamudi RK, Bulla LAJ. Insect Biochem Molec Biol. 2002;32:1025–1036. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

