Surface engineering of quantum dots for in vivo vascular imaging
- PMID: 17760416
- PMCID: PMC2853010
- DOI: 10.1021/bc070020r
Surface engineering of quantum dots for in vivo vascular imaging
Abstract
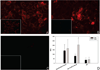
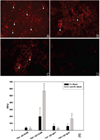
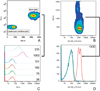
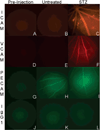
Quantum dot-antibody bioconjugates (QD-mAb) were synthesized incorporating PEG cross-linkers and Fc-shielding mAb fragments to increase in vivo circulation times and targeting efficiency. Microscopy of endothelial cell cultures incubated with QD-mAb directed against cell adhesion molecules (CAMs), when shielded to reduce Fc-mediated interactions, were more specific for their molecular targets. In vitro flow cytometry indicated that surface engineered QD-mAb labeled leukocyte subsets with minimal Fc-mediated binding. Nontargeted QD-mAb nanoparticles with Fc-blockade featured 64% (endothelial cells) and 53% (leukocytes) lower nonspecific binding than non-Fc-blocked nanoparticles. Spectrally distinct QD-mAb targeted to the cell adhesion molecules (CAMs) PECAM-1, ICAM-1, and VCAM-1 on the retinal endothelium in a rat model of diabetes were imaged in vivo using fluorescence angiography. Endogenously labeled circulating and adherent leukocyte subsets were imaged in rat models of diabetes and uveitis using QD-mAb targeted to RP-1 and CD45. Diabetic rats exhibited increased fluorescence in the retinal vasculature from QD bioconjugates to ICAM-1 and VCAM-1 but not PECAM-1. Both animal models exhibited leukocyte rolling and leukostasis in capillaries. Examination of retinal whole mounts prepared after in vivo imaging confirmed the fluorescence patterns seen in vivo. Comparison of the timecourse of retinal fluorescence from Fc-shielded and non-Fc-shielded bioconjugates indicated nonspecific uptake and increased clearance of the non-Fc-shielded QD-mAb. This combination of QD surface design elements offers a promising new in vivo approach to specifically label vascular cells and biomolecules of interest.
Figures



Similar articles
-
Integrin-mediated neutrophil adhesion and retinal leukostasis in diabetes.Invest Ophthalmol Vis Sci. 2000 Apr;41(5):1153-8. Invest Ophthalmol Vis Sci. 2000. PMID: 10752954
-
Endothelial cell E- and P-selectin and vascular cell adhesion molecule-1 function as signaling receptors.J Cell Biol. 1998 Sep 7;142(5):1381-91. doi: 10.1083/jcb.142.5.1381. J Cell Biol. 1998. PMID: 9732297 Free PMC article.
-
Leukocyte trafficking in experimental autoimmune uveitis: breakdown of blood-retinal barrier and upregulation of cellular adhesion molecules.Invest Ophthalmol Vis Sci. 2003 Jan;44(1):226-34. doi: 10.1167/iovs.01-1202. Invest Ophthalmol Vis Sci. 2003. PMID: 12506079
-
Polyethylene glycol–coated gold nanoshells conjugated with anti-VCAM-1 antibody.2013 Feb 28 [updated 2013 May 16]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2013 Feb 28 [updated 2013 May 16]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 23700638 Free Books & Documents. Review.
-
Anti-vascular cell adhesion molecule monoclonal antibody M/K-2.7 conjugated cross-linked iron oxide-Cy5.5 nanoparticles.2007 Oct 1 [updated 2007 Oct 29]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2007 Oct 1 [updated 2007 Oct 29]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 20641236 Free Books & Documents. Review.
Cited by
-
Dual-modal magnetic resonance and fluorescence imaging of atherosclerotic plaques in vivo using VCAM-1 targeted tobacco mosaic virus.Nano Lett. 2014 Mar 12;14(3):1551-8. doi: 10.1021/nl404816m. Epub 2014 Feb 17. Nano Lett. 2014. PMID: 24499194 Free PMC article.
-
Drug and gene delivery to the back of the eye: from bench to bedside.Invest Ophthalmol Vis Sci. 2014 Apr 28;55(4):2714-30. doi: 10.1167/iovs.13-13707. Invest Ophthalmol Vis Sci. 2014. PMID: 24777644 Free PMC article. Review. No abstract available.
-
Reduction of nanoparticle avidity enhances the selectivity of vascular targeting and PET detection of pulmonary inflammation.ACS Nano. 2013 Mar 26;7(3):2461-9. doi: 10.1021/nn305773f. Epub 2013 Feb 8. ACS Nano. 2013. PMID: 23383962 Free PMC article.
-
Recording the wild lives of immune cells.Sci Immunol. 2018 Sep 7;3(27):eaaq0491. doi: 10.1126/sciimmunol.aaq0491. Sci Immunol. 2018. PMID: 30194240 Free PMC article. Review.
-
Effects of Quantum Dot Labeling on Endothelial Progenitor Cell Function and Viability.Cell Med. 2010 Nov 5;1(2):105-12. doi: 10.3727/215517910X451603. eCollection 2010. Cell Med. 2010. PMID: 26966634 Free PMC article.
References
-
- Kaneider NC, Leger AJ, Kuliopulos A. Therapeutic targeting of molecules involved in leukocyte-endothelial cell interactions. FEBS J. 2006;273:4416–4424. - PubMed
-
- Frieri M. Asthma concepts in the new millennium: update in asthma pathophysiology. Allergy Asthma Proc. 2005;26:83–88. - PubMed
-
- Ulbrich H, Eriksson EE, Lindbom L. Leukocyte and endothelial cell adhesion molecules as targets for therapeutic interventions in inflammatory disease. Trends Pharmacol. Sci. 2003;24:640–647. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

