Abnormal oscillatory brain dynamics in schizophrenia: a sign of deviant communication in neural network?
- PMID: 17760978
- PMCID: PMC2034549
- DOI: 10.1186/1471-244X-7-44
Abnormal oscillatory brain dynamics in schizophrenia: a sign of deviant communication in neural network?
Abstract
Background: Slow waves in the delta (0.5-4 Hz) frequency range are indications of normal activity in sleep. In neurological disorders, focal electric and magnetic slow wave activity is generated in the vicinity of structural brain lesions. Initial studies, including our own, suggest that the distribution of the focal concentration of generators of slow waves (dipole density in the delta frequency band) also distinguishes patients with psychiatric disorders such as schizophrenia, affective disorders, and posttraumatic stress disorder.
Methods: The present study examined the distribution of focal slow wave activity (ASWA: abnormal slow wave activity) in 116 healthy subjects, 76 inpatients with schizophrenic or schizoaffective diagnoses and 42 inpatients with affective (ICD-10: F3) or neurotic/reactive (F4) diagnoses using a newly refined measure of dipole density. Based on 5-min resting magnetoencephalogram (MEG), sources of activity in the 1-4 Hz frequency band were determined by equivalent dipole fitting in anatomically defined cortical regions.
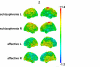
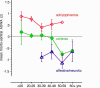
Results: Compared to healthy subjects the schizophrenia sample was characterized by significantly more intense slow wave activity, with maxima in frontal and central areas. In contrast, affective disorder patients exhibited less slow wave generators mainly in frontal and central regions when compared to healthy subjects and schizophrenia patients. In both samples, frontal ASWA were related to affective symptoms.
Conclusion: In schizophrenic patients, the regions of ASWA correspond to those identified for gray matter loss. This suggests that ASWA might be evaluated as a measure of altered neuronal network architecture and communication, which may mediate psychopathological signs.
Figures
Similar articles
-
Abnormal slow wave mapping (ASWAM)--A tool for the investigation of abnormal slow wave activity in the human brain.J Neurosci Methods. 2007 Jun 15;163(1):119-27. doi: 10.1016/j.jneumeth.2007.02.018. Epub 2007 Feb 23. J Neurosci Methods. 2007. PMID: 17395269
-
Source distribution of neuromagnetic slow waves and MEG-delta activity in schizophrenic patients.Biol Psychiatry. 2001 Jul 15;50(2):108-16. doi: 10.1016/s0006-3223(01)01122-2. Biol Psychiatry. 2001. PMID: 11526991 Clinical Trial.
-
Changes in cortical slow wave activity in healthy aging.Brain Imaging Behav. 2011 Sep;5(3):222-8. doi: 10.1007/s11682-011-9126-3. Brain Imaging Behav. 2011. PMID: 21698438
-
The visual scoring of sleep and arousal in infants and children.J Clin Sleep Med. 2007 Mar 15;3(2):201-40. J Clin Sleep Med. 2007. PMID: 17557427 Review.
-
Spontaneous low-frequency fluctuations in the BOLD signal in schizophrenic patients: anomalies in the default network.Schizophr Bull. 2007 Jul;33(4):1004-12. doi: 10.1093/schbul/sbm052. Epub 2007 Jun 7. Schizophr Bull. 2007. PMID: 17556752 Free PMC article. Review.
Cited by
-
Connectivity measures applied to human brain electrophysiological data.J Neurosci Methods. 2012 May 30;207(1):1-16. doi: 10.1016/j.jneumeth.2012.02.025. Epub 2012 Mar 16. J Neurosci Methods. 2012. PMID: 22426415 Free PMC article. Review.
-
Magnetoencephalography for Schizophrenia.Neuroimaging Clin N Am. 2020 May;30(2):205-216. doi: 10.1016/j.nic.2020.01.002. Epub 2020 Apr 9. Neuroimaging Clin N Am. 2020. PMID: 32336407 Free PMC article. Review.
-
Common Genomic and Proteomic Alterations Related to Disturbed Neural Oscillatory Activity in Schizophrenia.Int J Mol Sci. 2025 Aug 4;26(15):7514. doi: 10.3390/ijms26157514. Int J Mol Sci. 2025. PMID: 40806641 Free PMC article. Review.
-
Frontal slow-wave activity as a predictor of negative symptoms, cognition and functional capacity in schizophrenia.Br J Psychiatry. 2016 Feb;208(2):160-7. doi: 10.1192/bjp.bp.114.156075. Epub 2015 Jul 23. Br J Psychiatry. 2016. PMID: 26206861 Free PMC article.
-
Resting-state oscillatory activity in autism spectrum disorders.J Autism Dev Disord. 2012 Sep;42(9):1884-94. doi: 10.1007/s10803-011-1431-6. J Autism Dev Disord. 2012. PMID: 22207057 Free PMC article.
References
-
- Steriade M. Impact of network activities on neuronal properties in corticothalamic systems. J Neurophysiol. 2001;86:1–39. - PubMed
-
- Steriade M, Timofeev I. Neuronal plasticity in thalamocortical networks during sleep and waking oscillations. Neuron. 2003;89:1402–1413. - PubMed
-
- Walter G. The location of cerebral tumors by electroencephalography. Lancet. 1936;8:305–308. doi: 10.1016/S0140-6736(01)05173-X. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

