Microtiter plate monoclonal antibody epitope analysis of Ca2+- and Mg2+-induced conformational changes in troponin C
- PMID: 17761138
- PMCID: PMC2064003
- DOI: 10.1016/j.abb.2007.07.021
Microtiter plate monoclonal antibody epitope analysis of Ca2+- and Mg2+-induced conformational changes in troponin C
Abstract
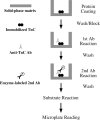
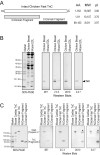
Spectroscopic methods such as circular dichroism and Förster resonance energy transfer are current approaches for monitoring protein conformational changes. Those analyses require special equipment and expertise. The need for fluorescence labeling of the protein may interfere with the native structure. We have developed a microtiter plate-based monoclonal antibody (mAb) epitope analysis to detect protein conformational changes in a high throughput manner. This method is based on the concept that the affinity of the antigen-binding site of an antibody for the specific antigenic epitope will change when the 3-D structure of the epitope changes. The effectiveness of this approach was demonstrated in the present study on troponin C (TnC), an allosteric protein in the Ca(2+) regulatory system of striated muscle. Using TnC purified by a highly effective rapid procedure and mAbs developed against epitopes in the N- and C-domains of TnC enzyme-linked immunosorbant assay (ELISA) clearly detected Ca(2+)-induced conformational changes in both the N-terminal regulatory domain and the C-terminal structural domain of TnC. On the other hand, Mg(2+)-binding to the C-domain of TnC resulted in a long-range effect on the N-domain conformation, indicating a functional significance of Ca(2+)-Mg(2+) exchange at the C-domain metal ion-binding sites. In addition to further understanding of the structure-function relationship of TnC, the data demonstrate that the mAb epitope analysis provides a simple high throughput method for monitoring 3-D structural changes in native proteins under physiological condition and has broad applications in protein structure-function relationship studies.
Figures


Similar articles
-
Calcium- and magnesium-dependent interactions between the C-terminus of troponin I and the N-terminal, regulatory domain of troponin C.Arch Biochem Biophys. 2001 Mar 15;387(2):243-9. doi: 10.1006/abbi.2000.2259. Arch Biochem Biophys. 2001. PMID: 11370847 Free PMC article.
-
Spectroscopic and ITC study of the conformational change upon Ca2+-binding in TnC C-lobe and TnI peptide complex from Akazara scallop striated muscle.Biochem Biophys Res Commun. 2008 Apr 25;369(1):109-14. doi: 10.1016/j.bbrc.2007.11.124. Epub 2007 Dec 3. Biochem Biophys Res Commun. 2008. PMID: 18054324
-
Ca2+-dependent interaction of the inhibitory region of troponin I with acidic residues in the N-terminal domain of troponin C.Biochim Biophys Acta. 1999 Mar 19;1430(2):214-21. doi: 10.1016/s0167-4838(99)00002-3. Biochim Biophys Acta. 1999. PMID: 10082949
-
The molecular switch in troponin C.Adv Exp Med Biol. 1993;332:117-23. doi: 10.1007/978-1-4615-2872-2_10. Adv Exp Med Biol. 1993. PMID: 8109324 Review.
-
[Conformational Fingerprinting Using Monoclonal Antibodies <br />(on the Example of Angiotensin I-Converting Enzyme-ACE)].Mol Biol (Mosk). 2017 Nov-Dec;51(6):1046-1061. doi: 10.7868/S0026898417060155. Mol Biol (Mosk). 2017. PMID: 29271967 Review. Russian.
Cited by
-
Early Divergence of the C-Terminal Variable Region of Troponin T Via a Pair of Mutually Exclusive Alternatively Spliced Exons Followed by a Selective Fixation in Vertebrate Heart.J Mol Evol. 2022 Dec;90(6):452-467. doi: 10.1007/s00239-022-10075-z. Epub 2022 Sep 28. J Mol Evol. 2022. PMID: 36171395 Free PMC article.
-
Nonmyofilament-associated troponin T fragments induce apoptosis.Am J Physiol Heart Circ Physiol. 2009 Jul;297(1):H283-92. doi: 10.1152/ajpheart.01200.2008. Epub 2009 Apr 24. Am J Physiol Heart Circ Physiol. 2009. PMID: 19395545 Free PMC article.
-
The glutamic acid-rich-long C-terminal extension of troponin T has a critical role in insect muscle functions.J Biol Chem. 2020 Mar 20;295(12):3794-3807. doi: 10.1074/jbc.RA119.012014. Epub 2020 Feb 5. J Biol Chem. 2020. PMID: 32024695 Free PMC article.
-
To investigate protein evolution by detecting suppressed epitope structures.J Mol Evol. 2009 May;68(5):448-60. doi: 10.1007/s00239-009-9202-0. Epub 2009 Apr 14. J Mol Evol. 2009. PMID: 19365646 Free PMC article.
-
A high-throughput solid-phase microplate protein-binding assay to investigate interactions between myofilament proteins.J Biomed Biotechnol. 2011;2011:421701. doi: 10.1155/2011/421701. Epub 2011 Nov 13. J Biomed Biotechnol. 2011. PMID: 22190850 Free PMC article.
References
-
- Kohler G, Milstein C. Nature. 1975;256:495–497. - PubMed
-
- Engvall E, Perlman P. Immunochemistry. 1971;8:871–874. - PubMed
-
- Parmacek MS, Leiden JM. Circulation. 1991;84:991–1003. - PubMed
-
- Gergely J, Grabarek Z, Tao T. Adv. Exp. Med. Biol. 1993;332:117–123. - PubMed
-
- Gordon AM, Homsher E, Regnier M. Physiol. Rev. 2000;80:853–924. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

