Stress-activated protein kinase-mediated down-regulation of the cell integrity pathway mitogen-activated protein kinase Pmk1p by protein phosphatases
- PMID: 17761528
- PMCID: PMC2043569
- DOI: 10.1091/mbc.e07-05-0484
Stress-activated protein kinase-mediated down-regulation of the cell integrity pathway mitogen-activated protein kinase Pmk1p by protein phosphatases
Abstract
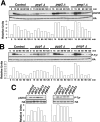
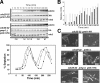
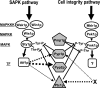
Fission yeast mitogen-activated protein kinase (MAPK) Pmk1p is involved in morphogenesis, cytokinesis, and ion homeostasis as part of the cell integrity pathway, and it becomes activated under multiple stresses, including hyper- or hypotonic conditions, glucose deprivation, cell wall-damaging compounds, and oxidative stress. The only protein phosphatase known to dephosphorylate and inactivate Pmk1p is Pmp1p. We show here that the stress-activated protein kinase (SAPK) pathway and its main effector, Sty1p MAPK, are essential for proper deactivation of Pmk1p under hypertonic stress in a process regulated by Atf1p transcription factor. We demonstrate that tyrosine phosphatases Pyp1p and Pyp2p, and serine/threonine phosphatase Ptc1p, that negatively regulate Sty1p activity and whose expression is dependent on Sty1p-Atf1p function, are involved in Pmk1p dephosphorylation under osmostress. Pyp1p and Ptc1p, in addition to Pmp1p, also control the basal level of MAPK Pmk1p activity in growing cells and associate with, and dephosphorylate Pmk1p both in vitro and in vivo. Our results with Ptc1p provide the first biochemical evidence for a PP2C-type phosphatase acting on more than one MAPK in yeast cells. Importantly, the SAPK-dependent down-regulation of Pmk1p through Pyp1p, Pyp2p, and Ptc1p was not complete, and Pyp1p and Ptc1p phosphatases are able to negatively regulate MAPK Pmk1p activity by an alternative regulatory mechanism. Our data also indicate that Pmk1p phosphorylation oscillates as a function of the cell cycle, peaking at cell separation during cytokinesis, and that Pmp1p phosphatase plays a main role in regulating this process.
Figures




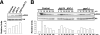


Similar articles
-
Activation of the cell integrity pathway is channelled through diverse signalling elements in fission yeast.Cell Signal. 2008 Apr;20(4):748-57. doi: 10.1016/j.cellsig.2007.12.017. Epub 2008 Jan 4. Cell Signal. 2008. PMID: 18255266
-
Heat-shock-induced activation of stress MAP kinase is regulated by threonine- and tyrosine-specific phosphatases.Genes Dev. 1999 Jul 1;13(13):1653-63. doi: 10.1101/gad.13.13.1653. Genes Dev. 1999. PMID: 10398679 Free PMC article.
-
Cold induces stress-activated protein kinase-mediated response in the fission yeast Schizosaccharomyces pombe.Eur J Biochem. 2002 Oct;269(20):5056-65. doi: 10.1046/j.1432-1033.2002.03214.x. Eur J Biochem. 2002. PMID: 12383265
-
The role of protein phosphatases in the regulation of mitogen and stress-activated protein kinases.Free Radic Res. 1999 Oct;31(4):341-9. doi: 10.1080/10715769900300911. Free Radic Res. 1999. PMID: 10517539 Review.
-
Regulation of stress-activated MAP kinase pathways during cell fate decisions.Nagoya J Med Sci. 2011 Feb;73(1-2):1-14. Nagoya J Med Sci. 2011. PMID: 21614932 Free PMC article. Review.
Cited by
-
The Role of the Cell Integrity Pathway in Septum Assembly in Yeast.J Fungi (Basel). 2021 Sep 6;7(9):729. doi: 10.3390/jof7090729. J Fungi (Basel). 2021. PMID: 34575767 Free PMC article. Review.
-
The Multiple Functions of Rho GTPases in Fission Yeasts.Cells. 2021 Jun 7;10(6):1422. doi: 10.3390/cells10061422. Cells. 2021. PMID: 34200466 Free PMC article. Review.
-
Role of the fission yeast cell integrity MAPK pathway in response to glucose limitation.BMC Microbiol. 2013 Feb 11;13:34. doi: 10.1186/1471-2180-13-34. BMC Microbiol. 2013. PMID: 23398982 Free PMC article.
-
Multiple protein kinases influence the redistribution of fission yeast Clp1/Cdc14 phosphatase upon genotoxic stress.Mol Biol Cell. 2012 Oct;23(20):4118-28. doi: 10.1091/mbc.E12-06-0475. Epub 2012 Aug 23. Mol Biol Cell. 2012. PMID: 22918952 Free PMC article.
-
Role of the RNA-binding protein Nrd1 and Pmk1 mitogen-activated protein kinase in the regulation of myosin mRNA stability in fission yeast.Mol Biol Cell. 2009 May;20(9):2473-85. doi: 10.1091/mbc.e08-09-0893. Epub 2009 Mar 11. Mol Biol Cell. 2009. PMID: 19279143 Free PMC article.
References
-
- Alfa C., Fantes P., Hyams J., Mcleod M., Warbrick E. New York: Cold Spring Harbor Laboratory Press; 1993. Experiments with Fission Yeast: A Laboratory Course Manual.
-
- Bahler J., Wu J. Q., Longtine M. S., Shah N. G., McKenzie A., 3rd, Steever A. B., Wach A., Philippsen P., Pringle J. R. Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast. 1998;14:943–951. - PubMed
-
- Bone N., Millar J. B., Toda T., Armstrong J. Regulated vacuole fusion and fission in Schizosaccharomyces pombe: an osmotic response dependent on MAP kinases. Curr. Biol. 1998;8:135–144. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

