Differential regulation of action potential firing in adult murine thalamocortical neurons by Kv3.2, Kv1, and SK potassium and N-type calcium channels
- PMID: 17761775
- PMCID: PMC2277158
- DOI: 10.1113/jphysiol.2007.141135
Differential regulation of action potential firing in adult murine thalamocortical neurons by Kv3.2, Kv1, and SK potassium and N-type calcium channels
Abstract
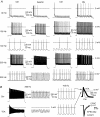
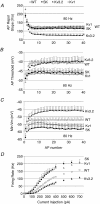
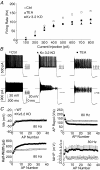
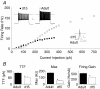
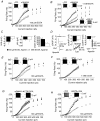
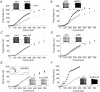
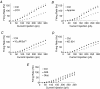
Sensory signals of widely differing dynamic range and intensity are transformed into a common firing rate code by thalamocortical neurons. While a great deal is known about the ionic currents, far less is known about the specific channel subtypes regulating thalamic firing rates. We hypothesized that different K(+) and Ca(2+) channel subtypes control different stimulus-response curve properties. To define the channels, we measured firing rate while pharmacologically or genetically modulating specific channel subtypes. Inhibiting Kv3.2 K(+) channels strongly suppressed maximum firing rate by impairing membrane potential repolarization, while playing no role in the firing response to threshold stimuli. By contrast, inhibiting Kv1 channels with alpha-dendrotoxin or maurotoxin strongly increased firing rates to threshold stimuli by reducing the membrane potential where action potentials fire (V(th)). Inhibiting SK Ca(2+)-activated K(+) channels with apamin robustly increased gain (slope of the stimulus-response curve) and maximum firing rate, with minimum effects on threshold responses. Inhibiting N-type Ca(2+) channels with omega-conotoxin GVIA or omega-conotoxin MVIIC partially mimicked apamin, while inhibiting L-type and P/Q-type Ca(2+) channels had small or no effects. EPSC-like current injections closely mimicked the results from tonic currents. Our results show that Kv3.2, Kv1, SK potassium and N-type calcium channels strongly regulate thalamic relay neuron sensory transmission and that each channel subtype controls a different stimulus-response curve property. Differential regulation of threshold, gain and maximum firing rate may help vary the stimulus-response properties across and within thalamic nuclei, normalize responses to diverse sensory inputs, and underlie sensory perception disorders.
Figures

Similar articles
-
Interactions between calcium channels and SK channels in midbrain dopamine neurons and their impact on pacemaker regularity: Contrasting roles of N- and L-type channels.Eur J Pharmacol. 2016 Oct 5;788:274-279. doi: 10.1016/j.ejphar.2016.06.046. Epub 2016 Jun 27. Eur J Pharmacol. 2016. PMID: 27364758
-
Calcium influx through N-type channels and activation of SK and TRP-like channels regulates tonic firing of neurons in rat paraventricular thalamus.J Neurophysiol. 2013 Nov;110(10):2450-64. doi: 10.1152/jn.00363.2013. Epub 2013 Sep 4. J Neurophysiol. 2013. PMID: 24004531
-
SK Channels Regulate Resting Properties and Signaling Reliability of a Developing Fast-Spiking Neuron.J Neurosci. 2017 Nov 1;37(44):10738-10747. doi: 10.1523/JNEUROSCI.1243-17.2017. Epub 2017 Oct 5. J Neurosci. 2017. PMID: 28982705 Free PMC article.
-
Molecular and cellular basis of small--and intermediate-conductance, calcium-activated potassium channel function in the brain.Cell Mol Life Sci. 2008 Oct;65(20):3196-217. doi: 10.1007/s00018-008-8216-x. Cell Mol Life Sci. 2008. PMID: 18597044 Free PMC article. Review.
-
Matching molecules to function: neuronal Ca2+-activated K+ channels and afterhyperpolarizations.Toxicon. 2004 Jun 15;43(8):933-49. doi: 10.1016/j.toxicon.2003.12.009. Toxicon. 2004. PMID: 15208027 Review.
Cited by
-
Modulation of Hyperpolarization-Activated Inward Current and Thalamic Activity Modes by Different Cyclic Nucleotides.Front Cell Neurosci. 2018 Oct 24;12:369. doi: 10.3389/fncel.2018.00369. eCollection 2018. Front Cell Neurosci. 2018. PMID: 30405353 Free PMC article.
-
Transient Hearing Loss Within a Critical Period Causes Persistent Changes to Cellular Properties in Adult Auditory Cortex.Cereb Cortex. 2015 Aug;25(8):2083-94. doi: 10.1093/cercor/bhu013. Epub 2014 Feb 18. Cereb Cortex. 2015. PMID: 24554724 Free PMC article.
-
Serotonin modulates multiple calcium current subtypes in commissural interneurons of the neonatal mouse.J Neurophysiol. 2012 Apr;107(8):2212-9. doi: 10.1152/jn.00768.2011. Epub 2012 Jan 25. J Neurophysiol. 2012. PMID: 22279189 Free PMC article.
-
The sleep relay--the role of the thalamus in central and decentral sleep regulation.Pflugers Arch. 2012 Jan;463(1):53-71. doi: 10.1007/s00424-011-1014-6. Epub 2011 Sep 13. Pflugers Arch. 2012. PMID: 21912835 Review.
-
Thalamic Kv 7 channels: pharmacological properties and activity control during noxious signal processing.Br J Pharmacol. 2015 Jun;172(12):3126-40. doi: 10.1111/bph.13113. Epub 2015 Apr 10. Br J Pharmacol. 2015. PMID: 25684311 Free PMC article.
References
-
- Abel HJ, Lee JCF, Callaway JC, Foehring RC. Relationships between intracellular calcium and afterhyperpolarizations in neocortical pyramial neurons. J Neurophysiol. 2004;91:324–335. - PubMed
-
- Atzori M, Lau D, Tansey EP, Chow A, Ozaita A, Rudy B, McBain CJ. H2 histamine receptor-phosphorylation of Kv3.2 modulates interneuron fast spiking. Nat Neurosci. 2000;3:791–798. - PubMed
-
- Bassett JP, Zugaro MB, Muir GM, Golob EJ, Muller RU, Taube JS. Passive movements of the head do not abolish anticipatory firing properties of head direction cells. J Neurophysiol. 2005;93:1304–1316. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

