Differential gene expression during terminal erythroid differentiation
- PMID: 17764892
- PMCID: PMC2205530
- DOI: 10.1016/j.ygeno.2007.06.010
Differential gene expression during terminal erythroid differentiation
Abstract
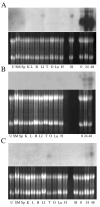
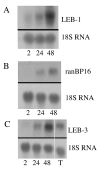
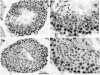
Terminal erythroid differentiation in mammals is the process whereby nucleated precursor cells accumulate erythroid-specific proteins such as hemoglobin, undergo extensive cellular and nuclear remodeling, and ultimately shed their nuclei to form reticulocytes, which then become mature erythrocytes in the circulation. Little is known about the mechanisms that enable erythroblasts to undergo such a transformation. We hypothesized that genes involved in these mechanisms were likely expressed at restricted times during the differentiation process and used differential display reverse transcriptase polymerase chain reaction as a first step in identifying such genes. We identified three differentially expressed cDNAs that we termed late erythroblast (LEB) 1-3. None of these cDNAs were previously identified as being expressed in erythroblasts and their patterns of expression indicated they are likely to be involved in the differentiation process. LEB-1 cDNA was derived from the gene A330102K04Rik (approved gene symbol Apoll1), and shares homology with members of the apolipoprotein L family in humans. LEB-3 cDNA was derived from the novel gene D930015E06Rik, that has no known function. LEB-2 cDNA was derived from the gene ranBP16 (approved gene symbol Xpo7), a nuclear exportin. D930015E06Rik mRNA is also strongly expressed in the testis and was localized to a region of the seminiferous tubule where secondary spermatocytes and early spermatids are found, suggesting a role for D930015E06Rik in spermatogenesis as well as terminal erythroid differentiation. We have thus identified three genes not previously described as being expressed in erythroblasts that could be relevant in elucidating mechanisms involved in terminal erythroid differentiation.
Figures

Similar articles
-
Expression of a large number of novel testis-specific genes during spermatogenesis coincides with the functional reorganization of the male germ cell.Int J Dev Biol. 1995 Oct;39(5):719-26. Int J Dev Biol. 1995. PMID: 8645556
-
A novel gene, RSD-3/HSD-3.1, encodes a meiotic-related protein expressed in rat and human testis.J Mol Med (Berl). 2003 Jun;81(6):380-7. doi: 10.1007/s00109-003-0434-y. Epub 2003 May 8. J Mol Med (Berl). 2003. PMID: 12736779
-
Complementary DNA cloning and characterization of rat spergen-2, a spermatogenic cell-specific gene 2 encoding a 56-kilodalton nuclear protein bearing ankyrin repeat motifs.Biol Reprod. 2003 Aug;69(2):421-9. doi: 10.1095/biolreprod.102.013987. Epub 2003 Mar 19. Biol Reprod. 2003. PMID: 12646494
-
Identification of Zfp393, a germ cell-specific gene encoding a novel zinc finger protein.Mech Dev. 2002 Oct;118(1-2):233-9. doi: 10.1016/s0925-4773(02)00258-7. Mech Dev. 2002. PMID: 12351194
-
Tsp57: a novel gene induced during a specific stage of spermatogenesis.Biol Reprod. 2004 Jan;70(1):106-13. doi: 10.1095/biolreprod.103.018465. Epub 2003 Sep 3. Biol Reprod. 2004. PMID: 12954732
Cited by
-
Importins and exportins in cellular differentiation.J Cell Mol Med. 2008 Oct;12(5B):1863-71. doi: 10.1111/j.1582-4934.2008.00437.x. Epub 2008 Jul 24. J Cell Mol Med. 2008. PMID: 18657223 Free PMC article. Review.
-
Global transcriptome analyses of human and murine terminal erythroid differentiation.Blood. 2014 May 29;123(22):3466-77. doi: 10.1182/blood-2014-01-548305. Epub 2014 Mar 17. Blood. 2014. PMID: 24637361 Free PMC article.
-
Cellular Zinc Deficiency Impairs Heme Biosynthesis in Developing Erythroid Progenitors.Nutrients. 2023 Jan 5;15(2):281. doi: 10.3390/nu15020281. Nutrients. 2023. PMID: 36678152 Free PMC article.
-
Proof of Gene Doping in a Mouse Model with a Human Erythropoietin Gene Transferred Using an Adenoviral Vector.Genes (Basel). 2021 Aug 16;12(8):1249. doi: 10.3390/genes12081249. Genes (Basel). 2021. PMID: 34440425 Free PMC article.
-
STRADalpha regulates LKB1 localization by blocking access to importin-alpha, and by association with Crm1 and exportin-7.Mol Biol Cell. 2008 Apr;19(4):1614-26. doi: 10.1091/mbc.e07-05-0454. Epub 2008 Feb 6. Mol Biol Cell. 2008. PMID: 18256292 Free PMC article.
References
-
- Dessypris EN. Erythropoiesis. In: Lee GR, Bithell TC, Foerster J, Athens JW, Lukens JN, editors. Wintrobe's Clinical Hematology. 9. Vol. 1. Lea & Febiger; Philadelphia: 1993.
-
- Lazarides E, Woods C. Biogenesis of the red blood cell membrane-skeleton and the control of erythroid morphogenesis. Annual Review of Cell Biology. 1989;5:427–52. - PubMed
-
- Koury MJ, Bondurant MC, Mueller TJ. The role of erythropoietin in the production of principal erythrocyte proteins other than hemoglobin during terminal erythroid differentiation. Journal of Cellular Physiology. 1986;126:259–65. - PubMed
-
- Nehls V, Drenckhahn D, Joshi R, Bennett V. Adducin in erythrocyte precursor cells of rats and humans: expression and compartmentalization. Blood. 1991;78:1692–6. - PubMed
-
- Hanspal M, Hanspal JS, Kalraiya R, Liu SC, Sahr KE, Howard D, Palek J. Asynchronous synthesis of membrane skeletal proteins during terminal maturation of murine erythroblasts. Blood. 1992a;80:530–9. - PubMed
Publication types
MeSH terms
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

