Effect of the structure of lipids favoring disordered domain formation on the stability of cholesterol-containing ordered domains (lipid rafts): identification of multiple raft-stabilization mechanisms
- PMID: 17766350
- PMCID: PMC2098711
- DOI: 10.1529/biophysj.107.114967
Effect of the structure of lipids favoring disordered domain formation on the stability of cholesterol-containing ordered domains (lipid rafts): identification of multiple raft-stabilization mechanisms
Abstract
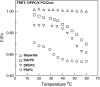
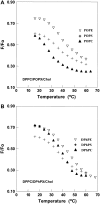
Despite the importance of lipid rafts, commonly defined as liquid-ordered domains rich in cholesterol and in lipids with high gel-to-fluid melting temperatures (T(m)), the rules for raft formation in membranes are not completely understood. Here, a fluorescence-quenching strategy was used to define how lipids with low T(m), which tend to form disordered fluid domains at physiological temperatures, can stabilize ordered domain formation by cholesterol and high-T(m) lipids (either sphingomyelin or dipalmitoylphosphatidylcholine). In bilayers containing mixtures of low-T(m) phosphatidylcholines, cholesterol, and high-T(m) lipid, the thermal stability of ordered domains decreased with the acyl-chain structure of low-T(m) lipids in the following order: diarachadonyl > diphytanoyl > 1-palmitoyl 2-docosahexenoyl = 1,2 dioleoyl = dimyristoleoyl = 1-palmitoyl, 2-oleoyl (PO). This shows that low-T(m) lipids with two acyl chains having very poor tight-packing propensities can stabilize ordered domain formation by high-T(m) lipids and cholesterol. The effect of headgroup structure was also studied. We found that even in the absence of high-T(m) lipids, mixtures of cholesterol with PO phosphatidylethanolamine (POPE) and PO phosphatidylserine (POPS) or with brain PE and brain PS showed a (borderline) tendency to form ordered domains. Because these lipids are abundant in the inner (cytofacial) leaflet of mammalian membranes, this raises the possibility that PE and PS could participate in inner-leaflet raft formation or stabilization. In bilayers containing ternary mixtures of PO lipids, cholesterol, and high-T(m) lipids, the thermal stability of ordered domains decreased with the polar headgroup structure of PO lipids in the order PE > PS > phosphatidylcholine (PC). Analogous experiments using diphytanoyl acyl chain lipids in place of PO acyl chain lipids showed that the stabilization of ordered lipid domains by acyl chain and headgroup structure was not additive. This implies that it is likely that there are two largely mutually exclusive mechanisms by which low-T(m) lipids can stabilize ordered domain formation by high-T(m) lipids and cholesterol: 1), by having structures resulting in immiscibility of low-T(m) and high-T(m) lipids, and 2), by having structures allowing them to pack tightly within ordered domains to a significant degree.
Figures

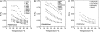
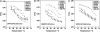


Similar articles
-
Effect of ceramide N-acyl chain and polar headgroup structure on the properties of ordered lipid domains (lipid rafts).Biochim Biophys Acta. 2007 Sep;1768(9):2205-12. doi: 10.1016/j.bbamem.2007.05.007. Epub 2007 May 13. Biochim Biophys Acta. 2007. PMID: 17574203 Free PMC article.
-
Transbilayer effects of raft-like lipid domains in asymmetric planar bilayers measured by single molecule tracking.Biophys J. 2006 Nov 1;91(9):3313-26. doi: 10.1529/biophysj.106.091421. Epub 2006 Aug 11. Biophys J. 2006. PMID: 16905614 Free PMC article.
-
Lipid rafts reconstituted in model membranes.Biophys J. 2001 Mar;80(3):1417-28. doi: 10.1016/S0006-3495(01)76114-0. Biophys J. 2001. PMID: 11222302 Free PMC article.
-
A lipid matrix model of membrane raft structure.Prog Lipid Res. 2010 Oct;49(4):390-406. doi: 10.1016/j.plipres.2010.05.002. Epub 2010 May 15. Prog Lipid Res. 2010. PMID: 20478335 Review.
-
The state of lipid rafts: from model membranes to cells.Annu Rev Biophys Biomol Struct. 2003;32:257-83. doi: 10.1146/annurev.biophys.32.110601.142439. Epub 2003 Jan 16. Annu Rev Biophys Biomol Struct. 2003. PMID: 12543707 Review.
Cited by
-
Investigation of channel-forming activity of polyene macrolide antibiotics in planar lipid bilayers in the presence of dipole modifiers.Acta Naturae. 2014 Oct;6(4):67-79. Acta Naturae. 2014. PMID: 25558397 Free PMC article.
-
Functional organization of the HIV lipid envelope.Sci Rep. 2016 Sep 28;6:34190. doi: 10.1038/srep34190. Sci Rep. 2016. PMID: 27678107 Free PMC article.
-
Using Sterol Substitution to Probe the Role of Membrane Domains in Membrane Functions.Lipids. 2015 Aug;50(8):721-34. doi: 10.1007/s11745-015-4007-y. Epub 2015 Mar 25. Lipids. 2015. PMID: 25804641 Free PMC article. Review.
-
Lipid Structure and Composition Control Consequences of Interleaflet Coupling in Asymmetric Vesicles.Biophys J. 2018 Aug 21;115(4):664-678. doi: 10.1016/j.bpj.2018.07.011. Epub 2018 Jul 19. Biophys J. 2018. PMID: 30082033 Free PMC article.
-
Lipid Chemical Structure Modulates the Disruptive Effects of Nanomaterials on Membrane Models.Langmuir. 2020 May 12;36(18):4923-4932. doi: 10.1021/acs.langmuir.0c00295. Epub 2020 Apr 30. Langmuir. 2020. PMID: 32312045 Free PMC article.
References
-
- Brown, D. A., and E. London. 1997. Structure of detergent-resistant membrane domains: does phase separation occur in biological membranes? Biochem. Biophys. Res. Commun. 240:1–7. - PubMed
-
- Brown, D. A., and E. London. 1998. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 14:111–136. - PubMed
-
- Almeida, P. F., W. L. Vaz, and T. E. Thompson. 1992. Lateral diffusion in the liquid phases of dimyristoylphosphatidylcholine/cholesterol lipid bilayers: a free volume analysis. Biochemistry. 31:6739–6747. - PubMed
-
- London, E. 2005. How principles of domain formation in model membranes may explain ambiguities concerning lipid raft formation in cells. Biochim. Biophys. Acta. 1746:203–220. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

