Dual targeting of antioxidant and metabolic enzymes to the mitochondrion and the apicoplast of Toxoplasma gondii
- PMID: 17784785
- PMCID: PMC1959373
- DOI: 10.1371/journal.ppat.0030115
Dual targeting of antioxidant and metabolic enzymes to the mitochondrion and the apicoplast of Toxoplasma gondii
Abstract
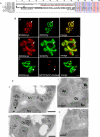
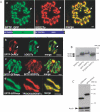
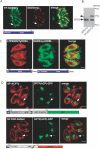
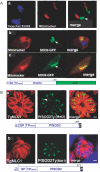
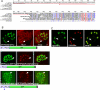
Toxoplasma gondii is an aerobic protozoan parasite that possesses mitochondrial antioxidant enzymes to safely dispose of oxygen radicals generated by cellular respiration and metabolism. As with most Apicomplexans, it also harbors a chloroplast-like organelle, the apicoplast, which hosts various biosynthetic pathways and requires antioxidant protection. Most apicoplast-resident proteins are encoded in the nuclear genome and are targeted to the organelle via a bipartite N-terminal targeting sequence. We show here that two antioxidant enzymes-a superoxide dismutase (TgSOD2) and a thioredoxin-dependent peroxidase (TgTPX1/2)-and an aconitase are dually targeted to both the apicoplast and the mitochondrion of T. gondii. In the case of TgSOD2, our results indicate that a single gene product is bimodally targeted due to an inconspicuous variation within the putative signal peptide of the organellar protein, which significantly alters its subcellular localization. Dual organellar targeting of proteins might occur frequently in Apicomplexans to serve important biological functions such as antioxidant protection and carbon metabolism.
Conflict of interest statement
Figures


Similar articles
-
An ambiguous N-terminus drives the dual targeting of an antioxidant protein Thioredoxin peroxidase (TgTPx1/2) to endosymbiotic organelles in Toxoplasma gondii.PeerJ. 2019 Jul 18;7:e7215. doi: 10.7717/peerj.7215. eCollection 2019. PeerJ. 2019. PMID: 31346496 Free PMC article.
-
Dually localised proteins found in both the apicoplast and mitochondrion utilize the Golgi-dependent pathway for apicoplast targeting in Toxoplasma gondii.Biol Cell. 2021 Jan;113(1):58-78. doi: 10.1111/boc.202000050. Epub 2020 Nov 23. Biol Cell. 2021. PMID: 33112425
-
The antioxidant systems in Toxoplasma gondii and the role of cytosolic catalase in defence against oxidative injury.Mol Microbiol. 2004 Jan;51(1):47-61. doi: 10.1046/j.1365-2958.2003.03823.x. Mol Microbiol. 2004. PMID: 14651610
-
Protein trafficking inside Toxoplasma gondii.Traffic. 2008 May;9(5):636-46. doi: 10.1111/j.1600-0854.2008.00713.x. Epub 2008 Mar 4. Traffic. 2008. PMID: 18331382 Review.
-
Towards a molecular architecture of the centrosome in Toxoplasma gondii.Cytoskeleton (Hoboken). 2017 Feb;74(2):55-71. doi: 10.1002/cm.21353. Epub 2017 Jan 11. Cytoskeleton (Hoboken). 2017. PMID: 28026138 Review.
Cited by
-
Mitochondrial metabolism of glucose and glutamine is required for intracellular growth of Toxoplasma gondii.Cell Host Microbe. 2012 Nov 15;12(5):682-92. doi: 10.1016/j.chom.2012.09.013. Cell Host Microbe. 2012. PMID: 23159057 Free PMC article.
-
Genetic screens reveal a central role for heme metabolism in artemisinin susceptibility.Nat Commun. 2020 Sep 23;11(1):4813. doi: 10.1038/s41467-020-18624-0. Nat Commun. 2020. PMID: 32968076 Free PMC article.
-
Differential contribution of two organelles of endosymbiotic origin to iron-sulfur cluster synthesis and overall fitness in Toxoplasma.PLoS Pathog. 2021 Nov 18;17(11):e1010096. doi: 10.1371/journal.ppat.1010096. eCollection 2021 Nov. PLoS Pathog. 2021. PMID: 34793583 Free PMC article.
-
The Toxoplasma gondii homolog of ATPase inhibitory factor 1 is critical for mitochondrial cristae maintenance and stress response.Mol Biol Cell. 2025 Jan 1;36(1):ar6. doi: 10.1091/mbc.E24-08-0344. Epub 2024 Nov 27. Mol Biol Cell. 2025. PMID: 39602296 Free PMC article.
-
Apicomplexan mitoribosome from highly fragmented rRNAs to a functional machine.Nat Commun. 2024 Dec 17;15(1):10689. doi: 10.1038/s41467-024-55033-z. Nat Commun. 2024. PMID: 39690155 Free PMC article.
References
-
- Ralph SA, Van Dooren GG, Waller RF, Crawford MJ, Fraunholz MJ, et al. Tropical infectious diseases: Metabolic maps and functions of the Plasmodium falciparum apicoplast. Nat Rev Microbiol. 2004;2:203–216. - PubMed
-
- Wilson RJ. Progress with parasite plastids. J Mol Biol. 2002;319:257–274. - PubMed
-
- Sato S, Clough B, Coates L, Wilson RJ. Enzymes for heme biosynthesis are found in both the mitochondrion and plastid of the malaria parasite Plasmodium falciparum . Protist. 2004;155:117–125. - PubMed
-
- Ellis KE, Clough B, Saldanha JW, Wilson RJ. Nifs and Sufs in malaria. Mol Microbiol. 2001;41:973–981. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

