Angiotensin receptors, autoimmunity, and preeclampsia
- PMID: 17785770
- PMCID: PMC3262172
- DOI: 10.4049/jimmunol.179.6.3391
Angiotensin receptors, autoimmunity, and preeclampsia
Abstract
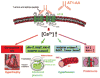
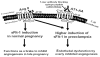
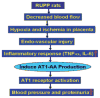
Preeclampsia is a pregnancy-induced hypertensive disorder that causes substantial maternal and fetal morbidity and mortality. Despite being a leading cause of maternal death and a major contributor to maternal and perinatal morbidity, the mechanisms responsible for the pathogenesis of preeclampsia are poorly understood. Recent studies indicate that women with preeclampsia have autoantibodies that activate the angiotensin receptor, AT1, and that autoantibody-mediated receptor activation contributes to pathophysiology associated with preeclampsia. The research reviewed here raises the intriguing possibility that preeclampsia may be a pregnancy-induced autoimmune disease.
Conflict of interest statement
The authors have no financial conflict of interest.
Figures
Similar articles
-
Is preeclampsia an autoimmune disease?Clin Immunol. 2009 Oct;133(1):1-12. doi: 10.1016/j.clim.2009.05.004. Epub 2009 Jun 5. Clin Immunol. 2009. PMID: 19501024 Free PMC article. Review.
-
Role of angiotensin II type I receptor agonistic autoantibodies (AT1-AA) in preeclampsia.Curr Opin Pharmacol. 2011 Apr;11(2):175-9. doi: 10.1016/j.coph.2011.01.003. Curr Opin Pharmacol. 2011. PMID: 21317038 Free PMC article. Review.
-
The Role of Agonistic Autoantibodies to the Angiotensin II Type 1 Receptor (AT1-AA) in Pathophysiology of Preeclampsia.Curr Pharm Biotechnol. 2018;19(10):781-785. doi: 10.2174/1389201019666180925121254. Curr Pharm Biotechnol. 2018. PMID: 30255752 Free PMC article. Review.
-
Angiotensin receptor agonistic autoantibodies and hypertension: preeclampsia and beyond.Circ Res. 2013 Jun 21;113(1):78-87. doi: 10.1161/CIRCRESAHA.113.300752. Circ Res. 2013. PMID: 23788505 Free PMC article. Review.
-
Lipoxin A4 suppresses angiotensin II type 1 receptor autoantibody in preeclampsia via modulating caspase-1.Cell Death Dis. 2020 Jan 30;11(1):78. doi: 10.1038/s41419-020-2281-y. Cell Death Dis. 2020. PMID: 32001671 Free PMC article.
Cited by
-
RAS in Pregnancy and Preeclampsia and Eclampsia.Int J Hypertens. 2012;2012:739274. doi: 10.1155/2012/739274. Epub 2012 Dec 30. Int J Hypertens. 2012. PMID: 23346385 Free PMC article.
-
Impact of Stress and Anxiety on Cardiovascular Health in Pregnancy: A Scoping Review.J Clin Med. 2025 Jan 30;14(3):909. doi: 10.3390/jcm14030909. J Clin Med. 2025. PMID: 39941580 Free PMC article. Review.
-
Maternal treatment with agonistic autoantibodies against type-1 angiotensin II receptor in late pregnancy increases apoptosis of myocardial cells and myocardial susceptibility to ischemia-reperfusion injury in offspring rats.PLoS One. 2013 Nov 21;8(11):e80709. doi: 10.1371/journal.pone.0080709. eCollection 2013. PLoS One. 2013. PMID: 24278308 Free PMC article.
-
Antibodies against AT1 receptors are associated with vascular endothelial and smooth muscle function impairment: protective effects of hydroxysafflor yellow A.PLoS One. 2013 Jun 24;8(6):e67020. doi: 10.1371/journal.pone.0067020. Print 2013. PLoS One. 2013. PMID: 23826187 Free PMC article.
-
Maternal Venous Hemodynamic Dysfunction in Proteinuric Gestational Hypertension: Evidence and Implications.J Clin Med. 2019 Mar 11;8(3):335. doi: 10.3390/jcm8030335. J Clin Med. 2019. PMID: 30862007 Free PMC article. Review.
References
-
- Redman CW, I, Sargent L. Latest advances in understanding preeclampsia. Science. 2005;308:1592–1594. - PubMed
-
- Roberts JM. Preeclampsia: what we know and what we do not know. Semin Perinatol. 2000;24:24–28. - PubMed
-
- Granger JP, Alexander BT, Llinas MT, Bennett WA, Khalil RA. Pathophysiology of hypertension during preeclampsia linking placental ischemia with endothelial dysfunction. Hypertension. 2001;38:718–722. - PubMed
-
- Gleicher N. Why much of the pathophysiology of preeclampsia-eclampsia must be of an autoimmune nature. Am J Obstet Gynecol. 2007;196:5.e1–7. - PubMed
-
- Sargent IL, Borzychowski AM, Redman CW. Immunoregulation in normal pregnancy and preeclampsia: an overview. Reprod Biomed Online. 2006;13:680–686. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

