The evolving biology and treatment of prostate cancer
- PMID: 17786228
- PMCID: PMC1952634
- DOI: 10.1172/JCI31791
The evolving biology and treatment of prostate cancer
Abstract
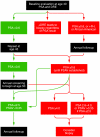
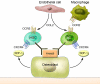
Since the effectiveness of androgen deprivation for treatment of advanced prostate cancer was first demonstrated, prevention strategies and medical therapies for prostate cancer have been based on understanding the biologic underpinnings of the disease. Prostate cancer treatment is one of the best examples of a systematic therapeutic approach to target not only the cancer cells themselves, but the microenvironment in which they are proliferating. As the population ages and prostate cancer prevalence increases, challenges remain in the diagnosis of clinically relevant prostate cancer as well as the management of the metastatic and androgen-independent metastatic disease states.
Figures




References
-
- American Cancer Society. 2007. Cancer facts and figures 2007. http://www.cancer.org/docroot/STT/content/STT_1x_Cancer_Facts__Figures_2....
-
- Scardino P.T., Weaver R., Hudson M.A. Early detection of prostate cancer. Hum. Pathol. 1992;23:211–222. - PubMed
-
- Bill-Axelson A., et al. Radical prostatectomy versus watchful waiting in early prostate cancer. . N. Engl. J. Med. 2005;352:1977–1984. - PubMed
-
- Etzioni R., et al. Overdiagnosis due to prostate-specific antigen screening: lessons from U.S. prostate cancer incidence trends. J. Natl. Cancer. Inst. 2002;94:981–990. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

