EGFR plays a pivotal role in the regulation of polyamine-dependent apoptosis in intestinal epithelial cells
- PMID: 17825525
- PMCID: PMC2699668
- DOI: 10.1016/j.cellsig.2007.08.001
EGFR plays a pivotal role in the regulation of polyamine-dependent apoptosis in intestinal epithelial cells
Abstract
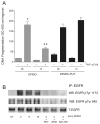
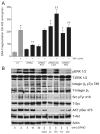
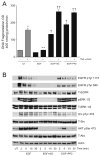
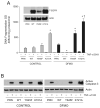
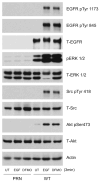
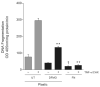
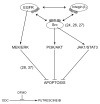
Intracellular polyamine synthesis is regulated by the enzyme ornithine decarboxylase (ODC), and its inhibition by alpha-difluromethylornithine (DFMO), confers resistance to apoptosis. We have previously shown that DFMO leads to the inhibition of de novo polyamine synthesis, which in turn rapidly activates Src, STAT3 and NF-kappaB via integrin beta3 in intestinal epithelial cells. One mechanism to explain these effects involves the activation of upstream growth factor receptors, such as the epidermal growth factor receptor (EGFR). We therefore hypothesized that EGFR phosphorylation regulates the early response to polyamine depletion. DFMO increased EGFR phosphorylation on tyrosine residues 1173 (pY1173) and 845 (pY845) within 5 min. Phosphorylation declined after 10 min and was prevented by the addition of exogenous putrescine to DFMO containing medium. Phosphorylation of EGFR was concomitant with the activation of ERK1/2. Pretreatment with either DFMO or EGF for 1 h protected cells from TNF-alpha/CHX-induced apoptosis. Exogenous addition of polyamines prevented the protective effect of DFMO. In addition, inhibition of integrin beta3 activity (with RGDS), Src activity (with PP2), or EGFR kinase activity (with AG1478), increased basal apoptosis and prevented protection conferred by either DFMO or EGF. Polyamine depletion failed to protect B82L fibroblasts lacking the EGFR (PRN) and PRN cells expressing either a kinase dead EGFR (K721A) or an EGFR (Y845F) mutant lacking the Src phosphorylation site. Conversely, expression of WT-EGFR (WT) restored the protective effect of polyamine depletion. Fibronectin activated the EGFR, Src, ERKs and protected cells from apoptosis. Taken together, our data indicate an essential role of EGFR kinase activity in MEK/ERK-mediated protection, which synergizes with integrin beta3 leading to Src-mediated protective responses in polyamine depleted cells.
Figures

Similar articles
-
Integrin beta3-mediated Src activation regulates apoptosis in IEC-6 cells via Akt and STAT3.Biochem J. 2006 Aug 1;397(3):437-47. doi: 10.1042/BJ20060256. Biochem J. 2006. PMID: 16669788 Free PMC article.
-
Effect of alpha-difluoromethyl-ornithine on the expression and function of the epidermal growth factor receptor in human breast epithelial cells in culture.Breast Cancer Res Treat. 2001 Jul;68(2):139-46. doi: 10.1023/a:1011923630307. Breast Cancer Res Treat. 2001. PMID: 11688517
-
Role of polyamines in p53-dependent apoptosis of intestinal epithelial cells.Cell Signal. 2009 Apr;21(4):509-22. doi: 10.1016/j.cellsig.2008.12.003. Epub 2008 Dec 24. Cell Signal. 2009. PMID: 19136059
-
Polyamines and nonmelanoma skin cancer.Toxicol Appl Pharmacol. 2007 Nov 1;224(3):249-56. doi: 10.1016/j.taap.2006.11.023. Epub 2006 Nov 29. Toxicol Appl Pharmacol. 2007. PMID: 17234230 Free PMC article. Review.
-
Disrupting polyamine homeostasis as a therapeutic strategy for neuroblastoma.Clin Cancer Res. 2009 Oct 1;15(19):5956-61. doi: 10.1158/1078-0432.CCR-08-3213. Epub 2009 Sep 29. Clin Cancer Res. 2009. PMID: 19789308 Free PMC article. Review.
Cited by
-
Dsg2 via Src-mediated transactivation shapes EGFR signaling towards cell adhesion.Cell Mol Life Sci. 2018 Nov;75(22):4251-4268. doi: 10.1007/s00018-018-2869-x. Epub 2018 Jul 6. Cell Mol Life Sci. 2018. PMID: 29980799 Free PMC article.
-
Elevated polyamines induce c-MYC overexpression by perturbing quadruplex-WC duplex equilibrium.Nucleic Acids Res. 2009 Jun;37(10):3321-31. doi: 10.1093/nar/gkp196. Epub 2009 Mar 26. Nucleic Acids Res. 2009. PMID: 19324889 Free PMC article.
-
Post-transcriptional regulation of MEK-1 by polyamines through the RNA-binding protein HuR modulating intestinal epithelial apoptosis.Biochem J. 2010 Feb 24;426(3):293-306. doi: 10.1042/BJ20091459. Biochem J. 2010. PMID: 20001965 Free PMC article.
-
Inhibition of Mdm2 sensitizes human retinal pigment epithelial cells to apoptosis.Invest Ophthalmol Vis Sci. 2011 May 1;52(6):3368-80. doi: 10.1167/iovs.10-6991. Invest Ophthalmol Vis Sci. 2011. PMID: 21345989 Free PMC article.
-
Transactivation of Epidermal Growth Factor Receptor by G Protein-Coupled Receptors: Recent Progress, Challenges and Future Research.Int J Mol Sci. 2016 Jan 12;17(1):95. doi: 10.3390/ijms17010095. Int J Mol Sci. 2016. PMID: 26771606 Free PMC article. Review.
References
-
- Cheng H, Leblond CP. Am J Anat. 1974;141:537. - PubMed
-
- Schmidt GH, Winton DJ, Ponder BAJ. Development. 1988;103:785. - PubMed
-
- Potten CS, Loeffler MA. J Theor Biol. 1987;127:381. - PubMed
-
- Wright NA, Irwin M. Cell Tissue Kinet. 1982;15:595. - PubMed
-
- Grossman J, Walther K, Artinger M, Kiessling S, Schlomerich J. Cell Growth Differ. 2001;12:147. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

