Cessation of renal morphogenesis in mice
- PMID: 17826763
- PMCID: PMC2075093
- DOI: 10.1016/j.ydbio.2007.08.021
Cessation of renal morphogenesis in mice
Abstract
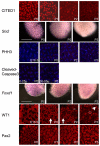
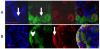
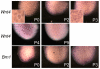
The kidney develops by cycles of ureteric bud branching and nephron formation. The cycles begin and are sustained by reciprocal inductive interactions and feedback between ureteric bud tips and the surrounding mesenchyme. Understanding how the cycles end is important because it controls nephron number. During the period when nephrogenesis ends in mice, we examined the morphology, gene expression, and function of the domains that control branching and nephrogenesis. We found that the nephrogenic mesenchyme, which is required for continued branching, was gone by the third postnatal day. This was associated with an accelerated rate of new nephron formation in the absence of apoptosis. At the same time, the tips of the ureteric bud branches lost the typical appearance of an ampulla and lost Wnt11 expression, consistent with the absence of the capping mesenchyme. Surprisingly, expression of Wnt9b, a gene necessary for mesenchyme induction, continued. We then tested the postnatal day three bud branch tip and showed that it maintained its ability both to promote survival of metanephric mesenchyme and to induce nephrogenesis in culture. These results suggest that the sequence of events leading to disruption of the cycle of branching morphogenesis and nephrogenesis began with the loss of mesenchyme that resulted from its conversion into nephrons.
Figures


References
-
- Carroll TJ, et al. Wnt9b plays a central role in the regulation of mesenchymal to epithelial transitions underlying organogenesis of the mammalian urogenital system. Dev Cell. 2005;9:283–92. - PubMed
-
- Chuang PT, McMahon AP. Branching morphogenesis of the lung: new molecular insights into an old problem. Trends Cell Biol. 2003;13:86–91. - PubMed
-
- Durbec P, et al. GDNF signalling through the Ret receptor tyrosine kinase. Nature. 1996;381:789–93. - PubMed
-
- Grieshammer U, et al. FGF8 is required for cell survival at distinct stages of nephrogenesis and for regulation of gene expression in nascent nephrons. Development. 2005;132:3847–57. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

