Regulation of nicotinic receptor trafficking by the transmembrane Golgi protein UNC-50
- PMID: 17853888
- PMCID: PMC2034668
- DOI: 10.1038/sj.emboj.7601858
Regulation of nicotinic receptor trafficking by the transmembrane Golgi protein UNC-50
Abstract
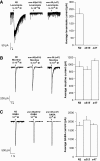
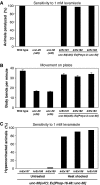
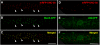
Nicotinic acetylcholine receptors (AChRs) are pentameric ligand-gated ion channels that mediate fast synaptic transmission at the neuromuscular junction (NMJ). After assembly in the endoplasmic reticulum (ER), AChRs must be transported to the plasma membrane through the secretory apparatus. Little is known about specific molecules that mediate this transport. Here we identify a gene that is required for subtype-specific trafficking of assembled nicotinic AChRs in Caenorhabditis elegans. unc-50 encodes an evolutionarily conserved integral membrane protein that localizes to the Golgi apparatus. In the absence of UNC-50, a subset of AChRs present in body-wall muscle are sorted to the lysosomal system and degraded. However, the trafficking of a second AChR type and of GABA ionotropic receptors expressed in the same muscle cells is not affected in unc-50 mutants. These results suggest that, in addition to ER quality control, assembled AChRs are sorted within the Golgi system by a mechanism that controls the amount of cell-surface AChRs in a subtype-specific way.
Figures





Similar articles
-
Structural determinants of alpha4beta2 nicotinic acetylcholine receptor trafficking.J Neurosci. 2005 Jul 13;25(28):6676-86. doi: 10.1523/JNEUROSCI.1079-05.2005. J Neurosci. 2005. PMID: 16014729 Free PMC article.
-
Biosynthesis of ionotropic acetylcholine receptors requires the evolutionarily conserved ER membrane complex.Proc Natl Acad Sci U S A. 2013 Mar 12;110(11):E1055-63. doi: 10.1073/pnas.1216154110. Epub 2013 Feb 19. Proc Natl Acad Sci U S A. 2013. PMID: 23431131 Free PMC article.
-
Determinants in the β and δ subunit cytoplasmic loop regulate Golgi trafficking and surface expression of the muscle acetylcholine receptor.J Biol Chem. 2014 Jan 3;289(1):203-14. doi: 10.1074/jbc.M113.502328. Epub 2013 Nov 15. J Biol Chem. 2014. PMID: 24240098 Free PMC article.
-
Protein networking: nicotinic acetylcholine receptors and their protein-protein-associations.Mol Cell Biochem. 2024 Jul;479(7):1627-1642. doi: 10.1007/s11010-024-05032-x. Epub 2024 May 21. Mol Cell Biochem. 2024. PMID: 38771378 Review.
-
Cholesterol effects on nicotinic acetylcholine receptor: cellular aspects.Subcell Biochem. 2010;51:467-87. doi: 10.1007/978-90-481-8622-8_17. Subcell Biochem. 2010. PMID: 20213555 Review.
Cited by
-
Levamisole receptors: a second awakening.Trends Parasitol. 2012 Jul;28(7):289-96. doi: 10.1016/j.pt.2012.04.003. Epub 2012 May 17. Trends Parasitol. 2012. PMID: 22607692 Free PMC article. Review.
-
Regulating the large Sec7 ARF guanine nucleotide exchange factors: the when, where and how of activation.Cell Mol Life Sci. 2014 Sep;71(18):3419-38. doi: 10.1007/s00018-014-1602-7. Epub 2014 Apr 13. Cell Mol Life Sci. 2014. PMID: 24728583 Free PMC article. Review.
-
Drug-efflux and target-site gene expression patterns in Haemonchus contortus larvae able to survive increasing concentrations of levamisole in vitro.Int J Parasitol Drugs Drug Resist. 2014 Mar 13;4(2):77-84. doi: 10.1016/j.ijpddr.2014.02.001. eCollection 2014 Aug. Int J Parasitol Drugs Drug Resist. 2014. PMID: 25057457 Free PMC article.
-
CRELD1 is an evolutionarily-conserved maturational enhancer of ionotropic acetylcholine receptors.Elife. 2018 Nov 7;7:e39649. doi: 10.7554/eLife.39649. Elife. 2018. PMID: 30407909 Free PMC article.
-
A Haploid Genetic Screen Identifies Heparan Sulfate Proteoglycans Supporting Rift Valley Fever Virus Infection.J Virol. 2015 Nov 18;90(3):1414-23. doi: 10.1128/JVI.02055-15. Print 2016 Feb 1. J Virol. 2015. PMID: 26581979 Free PMC article.
References
-
- Allan BB, Balch WE (1999) Protein sorting by directed maturation of Golgi compartments. Science 285: 63–66 - PubMed
-
- Arvan P, Zhao X, Ramos-Castaneda J, Chang A (2002) Secretory pathway quality control operating in Golgi, plasmalemmal, and endosomal systems. Traffic 3: 771–780 - PubMed
-
- Ballivet M, Alliod C, Bertrand S, Bertrand D (1996) Nicotinic acetylcholine receptors in the nematode Caenorhabditis elegans. J Mol Biol 258: 261–269 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

