Dual actin-bundling and protein kinase C-binding activities of fascin regulate carcinoma cell migration downstream of Rac and contribute to metastasis
- PMID: 17855511
- PMCID: PMC2043557
- DOI: 10.1091/mbc.e07-02-0157
Dual actin-bundling and protein kinase C-binding activities of fascin regulate carcinoma cell migration downstream of Rac and contribute to metastasis
Abstract
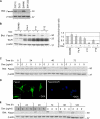
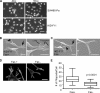
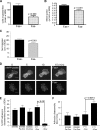
Recurrence of carcinomas due to cells that migrate away from the primary tumor is a major problem in cancer treatment. Immunohistochemical analyses of human carcinomas have consistently correlated up-regulation of the actin-bundling protein fascin with a clinically aggressive phenotype and poor prognosis. To understand the functional and mechanistic contributions of fascin, we undertook inducible short hairpin RNA (shRNA) knockdown of fascin in human colon carcinoma cells derived from an aggressive primary tumor. Fascin-depletion led to decreased numbers of filopodia and altered morphology of cell protrusions, decreased Rac-dependent migration on laminin, decreased turnover of focal adhesions, and, in vivo, decreased xenograft tumor development and metastasis. cDNA rescue of fascin shRNA-knockdown cells with wild-type green fluorescent protein-fascin or fascins mutated at the protein kinase C (PKC) phosphorylation site revealed that both the actin-bundling and active PKC-binding activities of fascin are required for the organization of filopodial protrusions, Rac-dependent migration, and tumor metastasis. Thus, fascin contributes to carcinoma migration and metastasis through dual pathways that impact on multiple subcellular structures needed for cell migration.
Figures



References
-
- Adams J. C. Roles of fascin in cell adhesion and motility. Curr. Opin. Cell Biol. 2004;16:590–596. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

