The thyroid hormone receptor-alpha (TRalpha) gene encoding TRalpha1 controls deoxyribonucleic acid damage-induced tissue repair
- PMID: 17872380
- PMCID: PMC5419624
- DOI: 10.1210/me.2007-0278
The thyroid hormone receptor-alpha (TRalpha) gene encoding TRalpha1 controls deoxyribonucleic acid damage-induced tissue repair
Abstract
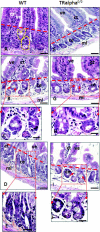
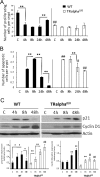
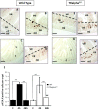
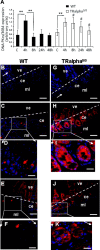
The thyroid hormone (TH) controls, via its nuclear receptor, TH receptor-alpha1 (TRalpha1), intestinal crypt cell proliferation in the mouse. In order to understand whether this receptor also plays a role in intestinal regeneration after DNA damage, we applied a protocol of gamma-ray irradiation and monitored cell proliferation and apoptosis at several time points. In wild-type mice, the dose of 8 Gy induced cell cycle arrest and apoptosis in intestinal crypts a few hours after irradiation. This phenomenon reverted 48 h after irradiation. TRalpha(0/0) mutant mice displayed a constant low level of proliferating cells and a high apoptosis rate during the period of study. At the molecular level, in TRalpha(0/0) animals we observed a delay in the p53 phosphorylation induced by DNA damage. In our search for the expression of the protein kinases responsible for p53 phosphorylation upon irradiation, we have focused on DNA-dependent protein kinase catalytic subunit (DNA-PKcs). The number of cells expressing DNA-PKcs in crypts remained high 48 h after irradiation, specifically in TRalpha mutants. Altogether, in TRalpha(0/0) animals the rate of apoptosis in crypt cells remained high, apparently due to an elevated number of cells still presenting DNA damage. In conclusion, the TRalpha gene plays a role in crypt cell homeostasis by regulating the rate of cell renewal and apoptosis induced by DNA damage.
Figures

References
-
- Brent GA 2000. Tissue-specific actions of thyroid hormone: insights from animal models. Rev Endocr Metab Disord 1:27–33 - PubMed
-
- Flamant F, Gauthier K, Samarut J 2007. Thyroid hormones signaling is getting more complex: STORMs are coming. Mol Endocrinol 21:321–333 - PubMed
-
- Plateroti M, Kress E, Samarut J 2007. Specific actions of thyroid hormones during gut development: insights from animal models. J Pediatr Endocrinol Metab 20:119–122
-
- Shi YB, Wong J, Puzianowska-Kuznicka M, Stolow MA 1996. Tadpole competence and tissue-specific temporal regulation of amphibian metamorphosis: roles of thyroid hormone and its receptors. Bioessays 18:391–399 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

