Fibroblast growth factor-2 regulates myocardial infarct repair: effects on cell proliferation, scar contraction, and ventricular function
- PMID: 17872976
- PMCID: PMC2043505
- DOI: 10.2353/ajpath.2007.070003
Fibroblast growth factor-2 regulates myocardial infarct repair: effects on cell proliferation, scar contraction, and ventricular function
Abstract
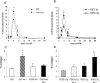
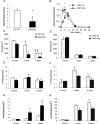
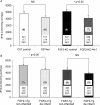
Fibroblast growth factor-2 (FGF2, bFGF) has been proposed to regulate wound healing and angiogenesis, but skin wound healing in FGF2-knockout (FGF2-KO) animals is only slightly delayed. To determine the role of FGF2 in myocardial infarct repair, we studied the evolution of left ventricular geometry, cell proliferation, matrix content, and cardiac function in mice lacking or overexpressing (FGF2-Tg) FGF2. Despite having no effect on initial infarct size, deletion of FGF2 resulted in reduced fibroblast proliferation and interstitial collagen deposition, decreased endothelial proliferation and vascular density, and decreased cardiomyocyte hypertrophy. Furthermore, FGF2-KO mice demonstrated a complete absence of scar contraction, resulting in increased final infarct size and marked increases in chamber size and infarct expansion. These deficits ultimately impaired left ventricular dP/dt compared with wild-type infarcted mice. Conversely, overexpression of FGF2 increased fibroblast proliferation and collagen deposition, accelerated endothelial proliferation, and enhanced cardiomyocyte hypertrophy after infarction. These changes curbed infarct expansion and preserved left ventricular function. Thus, FGF2 is an important regulator of cell proliferation, angiogenesis, collagen synthesis, myocyte hypertrophy, scar contraction, and, ultimately, left ventricular contractile function during infarct repair. FGF2 may be more important in healing of infarcts compared with skin wounds because of the mechanical stress under which infarcts heal.
Figures


References
-
- Boyle MP, Weisman HF. Limitation of infarct expansion and ventricular remodeling by late reperfusion. Study of time course and mechanism in a rat model. Circulation. 1993;88:2872–2883. - PubMed
-
- Detillieux KA, Sheikh F, Kardami E, Cattini PA. Biological activities of fibroblast growth factor-2 in the adult myocardium. Cardiovasc Res. 2003;57:8–19. - PubMed
-
- Kawai K, Suzuki S, Tabata Y, Ikada Y, Nishimura Y. Accelerated tissue regeneration through incorporation of basic fibroblast growth factor-impregnated gelatin microspheres into artificial dermis. Biomaterials. 2000;21:489–499. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

