PET studies of d-methamphetamine pharmacokinetics in primates: comparison with l-methamphetamine and ( --)-cocaine
- PMID: 17873134
- PMCID: PMC2732342
- DOI: 10.2967/jnumed.107.040279
PET studies of d-methamphetamine pharmacokinetics in primates: comparison with l-methamphetamine and ( --)-cocaine
Abstract
The methamphetamine molecule has a chiral center and exists as 2 enantiomers, d-methamphetamine (the more active enantiomer) and l-methamphetamine (the less active enantiomer). d-Methamphetamine is associated with more intense stimulant effects and higher abuse liability. The objective of this study was to measure the pharmacokinetics of d-methamphetamine for comparison with both l-methamphetamine and (-)-cocaine in the baboon brain and peripheral organs and to assess the saturability and pharmacologic specificity of binding.
Methods: d- and l-methamphetamine and (-)-cocaine were labeled with (11)C via alkylation of the norprecursors with (11)C-methyl iodide using literature methods. Six different baboons were studied in 11 PET sessions at which 2 radiotracer injections were administered 2-3 h apart to determine the distribution and kinetics of (11)C-d-methamphetamine in brain and peripheral organs. Saturability and pharmacologic specificity were assessed using pretreatment with d-methamphetamine, methylphenidate, and tetrabenazine. (11)C-d-Methamphetamine pharmacokinetics were compared with (11)C-l-methamphetamine and (11)C-(-)-cocaine in both brain and peripheral organs in the same animal.
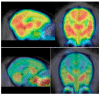
Results: (11)C-d- and l-methamphetamine both showed high uptake and widespread distribution in the brain. Pharmacokinetics did not differ between enantiomers, and the cerebellum peaked earlier and cleared more quickly than the striatum for both. (11)C-d-Methamphetamine distribution volume ratio was not substantially affected by pretreatment with methamphetamine, methylphenidate, or tetrabenazine. Both enantiomers showed rapid, high uptake and clearance in the heart and lungs and slower uptake and clearance in the liver and kidneys. A comparison of (11)C-d-methamphetamine and (11)C-(-)-cocaine showed that (11)C-d-methamphetamine peaked later in the brain than did (11)C-(-)-cocaine and cleared more slowly. The 2 drugs showed similar behavior in all peripheral organs examined except the kidneys and pancreas, which showed higher uptake for (11)C-d-methamphetamine.
Conclusion: Brain pharmacokinetics did not differ between d-and l-methamphetamine and thus cannot account for the more intense stimulant effects of d-methamphetamine. Lack of pharmacologic blockade by methamphetamine indicates that the PET image represents nonspecific binding, though the fact that methamphetamine is both a transporter substrate and an inhibitor may also play a role. A comparison of (11)C-d-methamphetamine and (11)C-(-)-cocaine in the same animal showed that the slower clearance of methamphetamine is likely to contribute to its previously reported longer-lasting stimulant effects relative to those of (-)-cocaine. High kidney uptake of d-methamphetamine or its labeled metabolites may account for the reported renal toxicity of d-methamphetamine in humans.
Figures




References
-
- Volkow ND, Chang L, Wang G-J, et al. Association of dopamine transporter reduction with psychomotor impairment in MAMP abusers. Am J Psychiatry. 2001;158:377–382. - PubMed
-
- Fitzmaurice PS, Tong J, Yazdanpanah M, Liu PP, Kalasinsky KS, Kish SJ. Levels of 4-hydroxynonenal and malondialdehyde are increased in brain of human chronic users of MAMP. J Pharmacol Exp Ther. 2006;319:703–709. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
