Identification of phosphotyrosine binding domain-containing proteins as novel downstream targets of the EphA8 signaling function
- PMID: 17875921
- PMCID: PMC2169194
- DOI: 10.1128/MCB.00794-07
Identification of phosphotyrosine binding domain-containing proteins as novel downstream targets of the EphA8 signaling function
Abstract
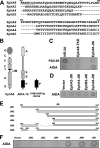
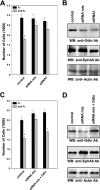
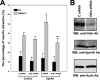
Eph receptors and ephrins have been implicated in a variety of cellular processes, including morphology and motility, because of their ability to modulate intricate signaling networks. Here we show that the phosphotyrosine binding (PTB) domain-containing proteins AIDA-1b and Odin are tightly associated with the EphA8 receptor in response to ligand stimulation. Both AIDA-1b and Odin belong to the ankyrin repeat and sterile alpha motif domain-containing (Anks) protein family. The PTB domain of Anks family proteins is crucial for their association with the juxtamembrane domain of EphA8, whereas EphA8 tyrosine kinase activity is not required for this protein-protein interaction. In addition, we found that Odin is a more physiologically relevant partner of EphA8 in mammalian cells. Interestingly, overexpression of the Odin PTB domain alone attenuated EphA8-mediated inhibition of cell migration in HEK293 cells, suggesting that it acts as a dominant-negative mutant of the endogenous Odin protein. More importantly, small interfering RNA-mediated Odin silencing significantly diminished ephrinA5-induced EphA8 signaling effects, which inhibit cell migration in HEK293 cells and retract growing neurites of Neuro2a cells. Taken together, our findings support a possible function for Anks family proteins as scaffolding proteins of the EphA8 signaling pathway.
Figures
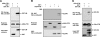





References
-
- Carter, N., T. Nakamoto, H. Hirai, and T. Hunter. 2002. EphrinA1-induced cytoskeletal re-organization requires FAK and p130(cas). Nat. Cell Biol. 4:565-573. - PubMed
-
- Choi, S., and S. Park. 1999. Phosphorylation at Tyr-838 in the kinase domain of EphA8 modulates Fyn binding to the Tyr-615 site by enhancing tyrosine kinase activity. Oncogene 18:5413-5422. - PubMed
-
- Dail, M., M. Richter, P. Godement, and E. B. Pasquale. 2006. Eph receptors inactivate R-Ras through different mechanisms to achieve cell repulsion. J. Cell Sci. 119:1244-1254. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
