Pharmacokinetics-pharmacodynamics of gatifloxacin in a lethal murine Bacillus anthracis inhalation infection model
- PMID: 17875992
- PMCID: PMC2167989
- DOI: 10.1128/AAC.00251-07
Pharmacokinetics-pharmacodynamics of gatifloxacin in a lethal murine Bacillus anthracis inhalation infection model
Abstract
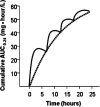
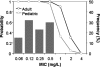
We determined the pharmacokinetic-pharmacodynamic (PK-PD) measure most predictive of gatifloxacin efficacy and the magnitude of this measure necessary for survival in a murine Bacillus anthracis inhalation infection model. We then used population pharmacokinetic models for gatifloxacin and simulation to identify dosing regimens with high probabilities of attaining exposures likely to be efficacious in adults and children. In this work, 6- to 8-week-old nonneutropenic female BALB/c mice received aerosol challenges of 50 to 75 50% lethal doses of B. anthracis (Ames strain, for which the gatifloxacin MIC is 0.125 mg/liter). Gatifloxacin was administered at 6- or 8-h intervals beginning 24 h postchallenge for 21 days, and dosing was designed to produce profiles mimicking fractionated concentration-time profiles for humans. Mice were evaluated daily for survival. Hill-type models were fitted to survival data. To identify potentially effective dosing regimens, adult and pediatric population pharmacokinetic models for gatifloxacin and Monte Carlo simulation were used to generate 5,000 individual patient exposure estimates. The ratio of the area under the concentration-time curve from 0 to 24 h (AUC(0-24)) to the MIC of the drug for the organism (AUC(0-24)/MIC ratio) was the PK-PD measure most predictive of survival (R(2) = 0.96). The 50% effective dose (ED(50)) and the ED(90) and ED(99) corresponded to AUC(0-24)/MIC ratios of 11.5, 15.8, and 30, respectively, where the maximum effect was 97% survival. Simulation results indicate that a daily gatifloxacin dose of 400 mg for adults and 10 mg/kg of body weight for children gives a 100% probability of attaining the PK-PD target (ED(99)). Sensitivity analyses suggest that the probability of PK-PD target attainment in adults and children is not affected by increases in MICs for strains of B. anthracis to levels as high as 0.5 mg/liter.
Figures
References
-
- Akaike, H. 1974. A new look at the statistical model identification. IEEE Trans. Automatic Control 19:716-723.
-
- Ambrose, P. G., S. M. Bhavnani, C. M. Rubino, A. Louie, T. Gumbo, A. Forrest, and G. L. Drusano. 2007. Pharmacokinetics-pharmacodynamics of antimicrobial therapy: it's not just for mice anymore. Clin. Infect. Dis. 44:79-86. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

