Update on neuropharmacological treatments for alcoholism: scientific basis and clinical findings
- PMID: 17880925
- PMCID: PMC2359153
- DOI: 10.1016/j.bcp.2007.08.005
Update on neuropharmacological treatments for alcoholism: scientific basis and clinical findings
Abstract
The past decade has seen an expansion of research and knowledge on pharmacotherapy for the treatment of alcohol dependence. The Food and Drug Administration (FDA)-approved medications naltrexone and acamprosate have shown mixed results in clinical trials. Oral naltrexone and naltrexone depot formulations have generally demonstrated efficacy at treating alcohol dependence, but their treatment effect size is small, and more research is needed to compare the effects of different doses on drinking outcome. Acamprosate has demonstrated efficacy for treating alcohol dependence in European trials, but with a small effect size. In U.S. trials, acamprosate has not proved to be efficacious. Research continues to explore which types of alcohol-dependent individual would benefit the most from treatment with naltrexone or acamprosate. The combination of the two medications demonstrated efficacy for treating alcohol dependence in one European study but not in a multi-site U.S. study. Another FDA-approved medication, disulfiram, is an aversive agent that does not diminish craving for alcohol. Disulfiram is most effective when given to those who are highly compliant or who are receiving their medication under supervision. Of the non-approved medications, topiramate is among the most promising, with a medium effect size in clinical trials. Another promising medication, baclofen, has shown efficacy in small trials. Serotonergic agents such as selective serotonin reuptake inhibitors and the serotonin-3 receptor antagonist, ondansetron, appear to be efficacious only among certain genetic subtypes of alcoholic. As neuroscientific research progresses, other promising medications, as well as medication combinations, for treating alcohol dependence continue to be explored.
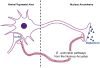
Figures



References
-
- U.S. Department of Health and Human Services. 10th Special Report to the US Congress on Alcohol and Health. Bethesda, MD: National Institute on Alcohol Abuse and Alcoholism; 2000.
-
- McLellan AT, Lewis DC, O’Brien CP, Kleber HD. Drug dependence, a chronic medical illness: implications for treatment, insurance, and outcomes evaluation. JAMA. 2000;284:1689–95. - PubMed
-
- Swift RM. Drug therapy for alcohol dependence. N Engl J Med. 1999;340:1482–90. - PubMed
-
- Finney JW, Hahn AC, Moos RH. The effectiveness of inpatient and outpatient treatment for alcohol abuse: the need to focus on mediators and moderators of setting effects. Addiction. 1996;91:1773–96. - PubMed
-
- Johnson BA, Ait-Daoud N, Akhtar FZ, Ma JZ. Oral topiramate reduces the consequences of drinking and improves the quality of life of alcohol-dependent individuals. Arch Gen Psychiatry. 2004;61:905–12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 5 R01 AA014628-03/AA/NIAAA NIH HHS/United States
- M01RR00847/RR/NCRR NIH HHS/United States
- R01 AA013964/AA/NIAAA NIH HHS/United States
- 7 R01 AA010522-12/AA/NIAAA NIH HHS/United States
- R01 AA012964/AA/NIAAA NIH HHS/United States
- 5 R01 AA012964-06/AA/NIAAA NIH HHS/United States
- U10 AA011776/AA/NIAAA NIH HHS/United States
- R01 AA010522/AA/NIAAA NIH HHS/United States
- M01 RR000847/RR/NCRR NIH HHS/United States
- R01 AA014628/AA/NIAAA NIH HHS/United States
- 5 R01 AA013964-03/AA/NIAAA NIH HHS/United States
- 7 U10 AA011776-10/AA/NIAAA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

