The mouse homeobox gene Noto regulates node morphogenesis, notochordal ciliogenesis, and left right patterning
- PMID: 17884984
- PMCID: PMC2000419
- DOI: 10.1073/pnas.0704344104
The mouse homeobox gene Noto regulates node morphogenesis, notochordal ciliogenesis, and left right patterning
Erratum in
- Proc Natl Acad Sci U S A. 2007 Oct 30;104(44):17554
Abstract
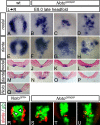
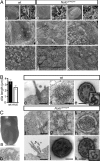
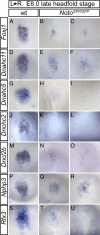
The mouse homeobox gene Noto represents the homologue of zebrafish floating head (flh) and is expressed in the organizer node and in the nascent notochord. Previous analyses suggested that Noto is required exclusively for the formation of the caudal part of the notochord. Here, we show that Noto is also essential for node morphogenesis, controlling ciliogenesis in the posterior notochord, and the establishment of laterality, whereas organizer functions in anterior-posterior patterning are apparently not compromised. In mutant embryos, left-right asymmetry of internal organs and expression of laterality markers was randomized. Mutant posterior notochord regions were variable in size and shape, cilia were shortened with highly irregular axonemal microtubuli, and basal bodies were, in part, located abnormally deep in the cytoplasm. The transcription factor Foxj1, which regulates the dynein gene Dnahc11 and is required for the correct anchoring of basal bodies in lung epithelial cells, was down-regulated in mutant nodes. Likewise, the transcription factor Rfx3, which regulates cilia growth, was not expressed in Noto mutants, and various other genes important for cilia function or assembly such as Dnahc5 and Nphp3 were down-regulated. Our results establish Noto as an essential regulator of node morphogenesis and ciliogenesis in the posterior notochord, and suggest Noto acts upstream of Foxj1 and Rfx3.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Morphogenesis of the node and notochord: the cellular basis for the establishment and maintenance of left-right asymmetry in the mouse.Dev Dyn. 2008 Dec;237(12):3464-76. doi: 10.1002/dvdy.21598. Dev Dyn. 2008. PMID: 18629866 Free PMC article. Review.
-
Differential regulation of node formation, nodal ciliogenesis and cilia positioning by Noto and Foxj1.Development. 2012 Apr;139(7):1276-84. doi: 10.1242/dev.072728. Epub 2012 Feb 22. Development. 2012. PMID: 22357932
-
A novel mammal-specific three partite enhancer element regulates node and notochord-specific Noto expression.PLoS One. 2012;7(10):e47785. doi: 10.1371/journal.pone.0047785. Epub 2012 Oct 22. PLoS One. 2012. PMID: 23110100 Free PMC article.
-
The mouse gene Noto is expressed in the tail bud and essential for its morphogenesis.Cells Tissues Organs. 2010;192(2):85-92. doi: 10.1159/000291015. Epub 2010 Feb 27. Cells Tissues Organs. 2010. PMID: 20197654
-
Of mice and men: dissecting the genetic pathway that controls left-right asymmetry in mice and humans.Am J Med Genet. 2000 Winter;97(4):258-70. Am J Med Genet. 2000. PMID: 11376437 Review.
Cited by
-
Gene Expression by Mouse Inner Ear Hair Cells during Development.J Neurosci. 2015 Apr 22;35(16):6366-80. doi: 10.1523/JNEUROSCI.5126-14.2015. J Neurosci. 2015. PMID: 25904789 Free PMC article.
-
THOC5/FMIP, an mRNA export TREX complex protein, is essential for hematopoietic primitive cell survival in vivo.BMC Biol. 2010 Jan 5;8:1. doi: 10.1186/1741-7007-8-1. BMC Biol. 2010. PMID: 20051105 Free PMC article.
-
Single-cell-resolved dynamics of chromatin architecture delineate cell and regulatory states in zebrafish embryos.Cell Genom. 2022 Jan 13;2(1):100083. doi: 10.1016/j.xgen.2021.100083. eCollection 2022 Jan 12. Cell Genom. 2022. PMID: 36777038 Free PMC article.
-
Vertebrate Left-Right Asymmetry: What Can Nodal Cascade Gene Expression Patterns Tell Us?J Cardiovasc Dev Dis. 2017 Dec 29;5(1):1. doi: 10.3390/jcdd5010001. J Cardiovasc Dev Dis. 2017. PMID: 29367579 Free PMC article. Review.
-
Morphogenesis of the node and notochord: the cellular basis for the establishment and maintenance of left-right asymmetry in the mouse.Dev Dyn. 2008 Dec;237(12):3464-76. doi: 10.1002/dvdy.21598. Dev Dyn. 2008. PMID: 18629866 Free PMC article. Review.
References
-
- Tam PPL, Gadd JM. In: Gastrulation: From Cells to Embryo. Stern C, editor. Cold Spring Harbor, NY: Cold Spring Harbor Lab Press; 2004. pp. 233–262.
-
- Camus A, Tam PP. Curr Top Dev Biol. 1999;45:117–153. - PubMed
-
- Viebahn C. Genesis. 2001;29:96–103. - PubMed
-
- Mackem S. In: Gastrulation: From Cells to Embryo. Stern CD, editor. Cold Spring Harbor, NY: Cold Spring Harbor Lab Press; 2004. pp. 599–607.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

