Positive feedback regulates switching of phosphate transporters in S. cerevisiae
- PMID: 17889672
- PMCID: PMC2034509
- DOI: 10.1016/j.molcel.2007.07.022
Positive feedback regulates switching of phosphate transporters in S. cerevisiae
Abstract
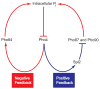
The regulation of transporters by nutrient-responsive signaling pathways allows cells to tailor nutrient uptake to environmental conditions. We investigated the role of feedback generated by transporter regulation in the budding yeast phosphate-responsive signal transduction (PHO) pathway. Cells starved for phosphate activate feedback loops that regulate high- and low-affinity phosphate transport. We determined that positive feedback is generated by PHO pathway-dependent upregulation of Spl2, a negative regulator of low-affinity phosphate uptake. The interplay of positive and negative feedback loops leads to bistability in phosphate transporter usage--individual cells express predominantly either low- or high-affinity transporters, both of which can yield similar phosphate uptake capacity. Cells lacking the high-affinity transporter, and associated negative feedback, exhibit phenotypes that arise from hysteresis due to unopposed positive feedback. In wild-type cells, population heterogeneity generated by feedback loops may provide a strategy for anticipating changes in environmental phosphate levels.
Figures


Similar articles
-
Uptake of selenite by Saccharomyces cerevisiae involves the high and low affinity orthophosphate transporters.J Biol Chem. 2010 Oct 15;285(42):32029-37. doi: 10.1074/jbc.M110.139865. Epub 2010 Aug 5. J Biol Chem. 2010. PMID: 20688911 Free PMC article.
-
The SPX domain of the yeast low-affinity phosphate transporter Pho90 regulates transport activity.EMBO Rep. 2009 Sep;10(9):1003-8. doi: 10.1038/embor.2009.105. Epub 2009 Jul 10. EMBO Rep. 2009. PMID: 19590579 Free PMC article.
-
Lack of 14-3-3 proteins in Saccharomyces cerevisiae results in cell-to-cell heterogeneity in the expression of Pho4-regulated genes SPL2 and PHO84.BMC Genomics. 2017 Sep 6;18(1):701. doi: 10.1186/s12864-017-4105-8. BMC Genomics. 2017. PMID: 28877665 Free PMC article.
-
Conservation of PHO pathway in ascomycetes and the role of Pho84.J Biosci. 2014 Jun;39(3):525-36. doi: 10.1007/s12038-014-9435-y. J Biosci. 2014. PMID: 24845516 Review.
-
Inorganic Phosphate and Sulfate Transport in S. cerevisiae.Adv Exp Med Biol. 2016;892:253-269. doi: 10.1007/978-3-319-25304-6_10. Adv Exp Med Biol. 2016. PMID: 26721277 Review.
Cited by
-
Evolution of reduced co-activator dependence led to target expansion of a starvation response pathway.Elife. 2017 May 9;6:e25157. doi: 10.7554/eLife.25157. Elife. 2017. PMID: 28485712 Free PMC article.
-
Heritable change caused by transient transcription errors.PLoS Genet. 2013 Jun;9(6):e1003595. doi: 10.1371/journal.pgen.1003595. Epub 2013 Jun 27. PLoS Genet. 2013. PMID: 23825966 Free PMC article.
-
A transcriptional activator from Rhizophagus irregularis regulates phosphate uptake and homeostasis in AM symbiosis during phosphorous starvation.Front Microbiol. 2023 Jan 20;13:1114089. doi: 10.3389/fmicb.2022.1114089. eCollection 2022. Front Microbiol. 2023. PMID: 36741887 Free PMC article.
-
Life in the midst of scarcity: adaptations to nutrient availability in Saccharomyces cerevisiae.Curr Genet. 2010 Feb;56(1):1-32. doi: 10.1007/s00294-009-0287-1. Curr Genet. 2010. PMID: 20054690 Review.
-
TGF-β1 promotes osteogenesis of mesenchymal stem cells via integrin mediated mechanical positive autoregulation.iScience. 2024 Jun 12;27(7):110262. doi: 10.1016/j.isci.2024.110262. eCollection 2024 Jul 19. iScience. 2024. PMID: 39021801 Free PMC article.
References
-
- Abelson PH. A potential phosphate crisis. Science. 1999;283:2015. - PubMed
-
- Acar M, Becskei A, van Oudenaarden A. Enhancement of cellular memory by reducing stochastic transitions. Nature. 2005;435:228–232. - PubMed
-
- Auesukaree C, Homma T, Kaneko Y, Harashima S. Transcriptional regulation of phosphate-responsive genes in low-affinity phosphate-transporter-defective mutants in Saccharomyces cerevisiae. Biochem Biophys Res Commun. 2003;306:843–850. - PubMed
-
- Barnes BR, Zierath JR. Role of AMP--activated protein kinase in the control of glucose homeostasis. Curr Mol Med. 2005;5:341–348. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

