Enhancement of L-selectin, but not P-selectin, bond formation frequency by convective flow
- PMID: 17890384
- PMCID: PMC2186251
- DOI: 10.1529/biophysj.106.098707
Enhancement of L-selectin, but not P-selectin, bond formation frequency by convective flow
Abstract
L-selectin-mediated leukocyte rolling has been proposed to require a high rate of bond formation compared to that of P-selectin to compensate for its much higher off-rate. To test this hypothesis, a microbead system was utilized to measure relative L-selectin and P-selectin bond formation rates on their common ligand P-selectin glycoprotein ligand-1 (PSGL-1) under shear flow. Using video microscopy, we tracked selectin-coated microbeads to detect the formation frequency of adhesive tether bonds. From velocity distributions of noninteracting and interacting microbeads, we observed that tether bond formation rates for P-selectin on PSGL-1 decreased with increasing wall shear stress, from 0.14 +/- 0.04 bonds/microm at 0.2 dyn/cm(2) to 0.014 +/- 0.003 bonds/microm at 1.0 dyn/cm(2). In contrast, L-selectin tether bond formation increased from 0.017 +/- 0.005 bonds/microm at 0.2 dyn/cm(2) to 0.031 +/- 0.005 bonds/microm at 1.0 dyn/cm(2). L-selectin tether bond formation rates appeared to be enhanced by convective transport, whereas P-selectin rates were inhibited. The transition force for the L-selectin catch-slip transition of 44 pN/bond agreed well with theoretical models (Pereverzev et al. 2005. Biophys. J. 89:1446-1454). Despite catch bond behavior, hydrodymanic shear thresholding was not detected with L-selectin beads rolling on PSGL-1. We speculate that shear flow generated compressive forces may enhance L-selectin bond formation relative to that of P-selectin and that L-selectin bonds with PSGL-1 may be tuned for the compressive forces characteristic of leukocyte-leukocyte collisions during secondary capture on the blood vessel wall. This is the first report, to our knowledge, comparing L-selectin and P-selectin bond formation frequencies in shear flow.
Figures



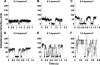



References
-
- Lawrence M.B. Selectin-carbohydrate interactions in shear flow. Curr. Opin. Chem. Biol. 1999;3:659–664. - PubMed
-
- Ley K., editor. Physiology of Inflammation. Oxford University Press; New York: 2001.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

