Serum withdrawal-induced accumulation of phosphoinositide 3-kinase lipids in differentiating 3T3-L6 myoblasts: distinct roles for Ship2 and PTEN
- PMID: 17893321
- PMCID: PMC2169165
- DOI: 10.1128/MCB.00756-07
Serum withdrawal-induced accumulation of phosphoinositide 3-kinase lipids in differentiating 3T3-L6 myoblasts: distinct roles for Ship2 and PTEN
Abstract
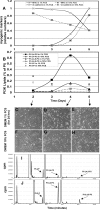
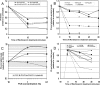
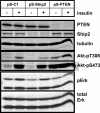
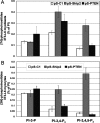
Phosphoinositide 3-kinase (PI3K) activation and synthesis of phosphatidylinositol-3,4-bisphosphate (PI-3,4-P2) and phosphatidylinositol-3,4,5-trisphosphate (PI-3,4,5-P3) lipids mediate growth factor signaling that leads to cell proliferation, migration, and survival. PI3K-dependent activation of Akt is critical for myoblast differentiation induced by serum withdrawal, suggesting that in these cells PI3K signaling is activated in an unconventional manner. Here we investigate the mechanisms by which PI3K signaling and Akt are regulated during myogenesis. We report that PI-3,4-P2 and PI-3,4,5-P3 accumulated in the plasma membranes of serum-starved 3T3-L6 myoblasts due to de novo synthesis and increased lipid stability. Surprisingly, only newly synthesized lipids were capable of activating Akt. Knockdown of the lipid phosphatase PTEN moderately increased PI3K lipids but significantly increased Akt phosphorylation and promoted myoblast differentiation. Knockdown of the lipid phosphatase Ship2, on the other hand, dramatically increased the steady-state levels of PI-3,4,5-P3 but did not affect Akt phosphorylation and increased apoptotic cell death. Together, these results reveal the existence of two distinct pools of PI3K lipids in differentiating 3T3-L6 myoblasts: a pool of nascent lipids that is mainly dephosphorylated by PTEN and is capable of activating Akt and promoting myoblast differentiation and a stable pool that is dephosphorylated by Ship2 and is unable to activate Akt.
Figures

Similar articles
-
PTEN, but not SHIP2, suppresses insulin signaling through the phosphatidylinositol 3-kinase/Akt pathway in 3T3-L1 adipocytes.J Biol Chem. 2005 Jun 10;280(23):22523-9. doi: 10.1074/jbc.M501949200. Epub 2005 Apr 11. J Biol Chem. 2005. PMID: 15824124
-
Imatinib mesilate-induced phosphatidylinositol 3-kinase signalling and improved survival in insulin-producing cells: role of Src homology 2-containing inositol 5'-phosphatase interaction with c-Abl.Diabetologia. 2013 Jun;56(6):1327-38. doi: 10.1007/s00125-013-2868-2. Epub 2013 Mar 5. Diabetologia. 2013. PMID: 23462796
-
PTEN and Other PtdIns(3,4,5)P3 Lipid Phosphatases in Breast Cancer.Int J Mol Sci. 2020 Dec 2;21(23):9189. doi: 10.3390/ijms21239189. Int J Mol Sci. 2020. PMID: 33276499 Free PMC article. Review.
-
Overexpression of SH2-containing inositol phosphatase 2 results in negative regulation of insulin-induced metabolic actions in 3T3-L1 adipocytes via its 5'-phosphatase catalytic activity.Mol Cell Biol. 2001 Mar;21(5):1633-46. doi: 10.1128/MCB.21.5.1633-1646.2001. Mol Cell Biol. 2001. PMID: 11238900 Free PMC article.
-
Phosphatidylinositol (3,4) bisphosphate-specific phosphatases and effector proteins: A distinct branch of PI3K signaling.Cell Signal. 2015 Sep;27(9):1789-98. doi: 10.1016/j.cellsig.2015.05.013. Epub 2015 May 27. Cell Signal. 2015. PMID: 26022180 Review.
Cited by
-
Role of phosphatidylinositol 3,4,5-trisphosphate (PIP3) 5-phosphatase skeletal muscle- and kidney-enriched inositol polyphosphate phosphatase (SKIP) in myoblast differentiation.J Biol Chem. 2012 Sep 7;287(37):31330-41. doi: 10.1074/jbc.M112.388785. Epub 2012 Jul 19. J Biol Chem. 2012. PMID: 22815484 Free PMC article.
-
The impact of phosphoinositide 5-phosphatases on phosphoinositides in cell function and human disease.J Lipid Res. 2019 Feb;60(2):276-286. doi: 10.1194/jlr.R087908. Epub 2018 Sep 7. J Lipid Res. 2019. PMID: 30194087 Free PMC article. Review.
-
Regulation of insulin signaling and glucose transporter 4 (GLUT4) exocytosis by phosphatidylinositol 3,4,5-trisphosphate (PIP3) phosphatase, skeletal muscle, and kidney enriched inositol polyphosphate phosphatase (SKIP).J Biol Chem. 2012 Mar 2;287(10):6991-9. doi: 10.1074/jbc.M111.335539. Epub 2012 Jan 15. J Biol Chem. 2012. PMID: 22247557 Free PMC article.
-
Therapeutic potential of SH2 domain-containing inositol-5'-phosphatase 1 (SHIP1) and SHIP2 inhibition in cancer.Mol Med. 2012 Feb 10;18(1):65-75. doi: 10.2119/molmed.2011.00178. Mol Med. 2012. PMID: 22033675 Free PMC article.
-
Targeting Plasmodium PI(4)K to eliminate malaria.Nature. 2013 Dec 12;504(7479):248-253. doi: 10.1038/nature12782. Epub 2013 Nov 27. Nature. 2013. PMID: 24284631 Free PMC article.
References
-
- Alessi, D. R., S. R. James, C. P. Downes, A. B. Holmes, P. R. Gaffney, C. B. Reese, and P. Cohen. 1997. Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Balpha. Curr. Biol. 7:261-269. - PubMed
-
- Blero, D., J. Zhang, X. Pesesse, B. Payrastre, J. E. Dumont, S. Shurmans, and C. Erneux. 2005. Phosphatidylinositol 3,4,5-trisphosphate modulation in Ship2 deficient mouse embryonic fibroblast. FEBS J. 272:2512-2522. - PubMed
-
- Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248-254. - PubMed
-
- Cantley, L. C. 2002. The phosphoinositide 3-kinase pathway. Science 296:1655-1657. - PubMed
-
- Clement, S., U. Krause, F. Desmedt, J. F. Tanti, J. Behrends, X. Pesesse, T. Sasaki, J. Penninger, M. Doherty, W. Malaisse, J. E. Dumont, Y. Le Marchand-Brustel, C. Erneux, L. Hue, and S. Schurmans. 2001. The lipid phosphatase SHIP2 controls insulin sensitivity. Nature 409:92-97. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
