Urodele p53 tolerates amino acid changes found in p53 variants linked to human cancer
- PMID: 17903248
- PMCID: PMC2072957
- DOI: 10.1186/1471-2148-7-180
Urodele p53 tolerates amino acid changes found in p53 variants linked to human cancer
Abstract
Background: Urodele amphibians like the axolotl are unique among vertebrates in their ability to regenerate and their resistance to develop cancers. It is unknown whether these traits are linked at the molecular level.
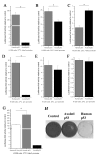
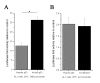
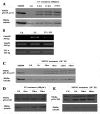
Results: Blocking p53 signaling in axolotls using the p53 inhibitor, pifithrin-alpha, inhibited limb regeneration and the expression of p53 target genes such as Mdm2 and Gadd45, suggesting a link between tumor suppression and regeneration. To understand this relationship we cloned the p53 gene from axolotl. When comparing its sequence with p53 from other organisms, and more specifically human we observed multiple amino acids changes found in human tumors. Phylogenetic analysis of p53 protein sequences from various species is in general agreement with standard vertebrate phylogeny; however, both mice-like rodents and teleost fishes are fast evolving. This leads to long branch attraction resulting in an artefactual basal emergence of these groups in the phylogenetic tree. It is tempting to assume a correlation between certain life style traits (e.g. lifespan) and the evolutionary rate of the corresponding p53 sequences. Functional assays of the axolotl p53 in human or axolotl cells using p53 promoter reporters demonstrated a temperature sensitivity (ts), which was further confirmed by performing colony assays at 37 degrees C. In addition, axolotl p53 was capable of efficient transactivation at the Hmd2 promoter but has moderate activity at the p21 promoter. Endogenous axolotl p53 was activated following UV irradiation (100 j/m2) or treatment with an alkylating agent as measured using serine 15 phosphorylation and the expression of the endogenous p53 target Gadd45.
Conclusion: Urodele p53 may play a role in regeneration and has evolved to contain multiple amino acid changes predicted to render the human protein defective in tumor suppression. Some of these mutations were probably selected to maintain p53 activity at low temperature. However, other significant changes in the axolotl proteins may play more subtle roles on p53 functions, including DNA binding and promoter specificity and could represent useful adaptations to ensure p53 activity and tumor suppression in animals able to regenerate or subject to large variations in oxygen levels or temperature.
Figures




Similar articles
-
The axolotl limb: a model for bone development, regeneration and fracture healing.Bone. 2007 Jan;40(1):45-56. doi: 10.1016/j.bone.2006.07.005. Epub 2006 Aug 21. Bone. 2007. PMID: 16920050
-
Novel human p53 mutations that are toxic to yeast can enhance transactivation of specific promoters and reactivate tumor p53 mutants.Oncogene. 2001 Jun 7;20(26):3409-19. doi: 10.1038/sj.onc.1204457. Oncogene. 2001. PMID: 11423991
-
P53 in blind subterranean mole rats--loss-of-function versus gain-of-function activities on newly cloned Spalax target genes.Oncogene. 2007 Apr 12;26(17):2507-12. doi: 10.1038/sj.onc.1210045. Epub 2006 Oct 16. Oncogene. 2007. PMID: 17043642
-
Structure of MHC class I and class II cDNAs and possible immunodeficiency linked to class II expression in the Mexican axolotl.Immunol Rev. 1998 Dec;166:259-77. doi: 10.1111/j.1600-065x.1998.tb01268.x. Immunol Rev. 1998. PMID: 9914918 Review.
-
Vertebrate cardiac regeneration: evolutionary and developmental perspectives.Cell Regen. 2021 Mar 1;10(1):6. doi: 10.1186/s13619-020-00068-y. Cell Regen. 2021. PMID: 33644818 Free PMC article. Review.
Cited by
-
Eya2 promotes cell cycle progression by regulating DNA damage response during vertebrate limb regeneration.Elife. 2020 Mar 6;9:e51217. doi: 10.7554/eLife.51217. Elife. 2020. PMID: 32142407 Free PMC article.
-
The Meis homeoprotein regulates the axolotl Prod 1 promoter during limb regeneration.Gene. 2011 Sep 15;484(1-2):69-74. doi: 10.1016/j.gene.2011.06.003. Epub 2011 Jun 12. Gene. 2011. PMID: 21684325 Free PMC article.
-
Functional convergence of signalling by GPI-anchored and anchorless forms of a salamander protein implicated in limb regeneration.J Cell Sci. 2011 Jan 1;124(Pt 1):47-56. doi: 10.1242/jcs.076331. Epub 2010 Nov 30. J Cell Sci. 2011. PMID: 21118959 Free PMC article.
-
Adrenergic signaling coordinates distant and local responses to amputation in axolotl.bioRxiv [Preprint]. 2025 Jul 24:2021.12.29.474455. doi: 10.1101/2021.12.29.474455. bioRxiv. 2025. PMID: 40777403 Free PMC article. Preprint.
-
Tumor suppressors: enhancers or suppressors of regeneration?Development. 2013 Jun;140(12):2502-12. doi: 10.1242/dev.084210. Development. 2013. PMID: 23715544 Free PMC article.
References
-
- Giaccia AJ, Kastan MB. The complexity of p53 modulation: emerging patterns from divergent signals. Genes Dev. 1998;12:2973–2983. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

