In vivo genetic profiling and cellular localization of apelin reveals a hypoxia-sensitive, endothelial-centered pathway activated in ischemic heart failure
- PMID: 17906101
- PMCID: PMC2570026
- DOI: 10.1152/ajpheart.00935.2007
In vivo genetic profiling and cellular localization of apelin reveals a hypoxia-sensitive, endothelial-centered pathway activated in ischemic heart failure
Abstract
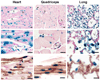
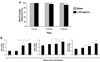
Signaling by the peptide ligand apelin and its cognate G protein-coupled receptor APJ has a potent inotropic effect on cardiac contractility and modulates systemic vascular resistance through nitric oxide-dependent signaling. In addition, there is evidence for counterregulation of the angiotensin and vasopressin pathways. Regulatory stimuli of the apelin-APJ pathway are of obvious importance but remain to be elucidated. To better understand the physiological response of apelin-APJ to disease states such as heart failure and to elucidate the mechanism by which such a response might occur, we have used the murine model of left anterior descending coronary artery ligation-induced ischemic cardiac failure. To identify the key cells responsible for modulation and production of apelin in vivo, we have created a novel apelin-lacZ reporter mouse. Data from these studies demonstrate that apelin and APJ are upregulated in the heart and skeletal muscle following myocardial injury and suggest that apelin expression remains restricted to the endothelium. In cardiac failure, endothelial apelin expression correlates with other hypoxia-responsive genes, and in healthy animals both apelin and APJ are markedly upregulated in various tissues following systemic hypoxic exposure. Experiments with cultured endothelial cells in vitro show apelin mRNA and protein levels to be increased by hypoxia, through a hypoxia-inducible factor-mediated pathway. These studies suggest that apelin-expressing endothelial cells respond to conditions associated with heart failure, possibly including local tissue hypoxia, and modulate apelin-APJ expression to regulate cardiovascular homeostasis. The apelin-APJ pathway may thus provide a mechanism for systemic endothelial monitoring of tissue perfusion and adaptive regulation of cardiovascular function.
Figures





Similar articles
-
Hypoxia-induced apelin expression regulates endothelial cell proliferation and regenerative angiogenesis.Circ Res. 2008 Aug 15;103(4):432-40. doi: 10.1161/CIRCRESAHA.108.179333. Epub 2008 Jul 10. Circ Res. 2008. PMID: 18617693
-
Disruption of the apelin-APJ system worsens hypoxia-induced pulmonary hypertension.Arterioscler Thromb Vasc Biol. 2011 Apr;31(4):814-20. doi: 10.1161/ATVBAHA.110.219980. Epub 2011 Jan 13. Arterioscler Thromb Vasc Biol. 2011. PMID: 21233449 Free PMC article.
-
Apelin/APJ signaling promotes hypoxia-induced proliferation of endothelial progenitor cells via phosphoinositide-3 kinase/Akt signaling.Mol Med Rep. 2015 Sep;12(3):3829-3834. doi: 10.3892/mmr.2015.3866. Epub 2015 May 27. Mol Med Rep. 2015. PMID: 26018184
-
Apelin/APJ signaling in hypoxia-related diseases.Clin Chim Acta. 2015 Dec 7;451(Pt B):191-8. doi: 10.1016/j.cca.2015.09.029. Epub 2015 Oct 3. Clin Chim Acta. 2015. PMID: 26436483 Review.
-
Apelin and its receptor APJ in cardiovascular diseases.Clin Chim Acta. 2014 Jan 20;428:1-8. doi: 10.1016/j.cca.2013.09.001. Epub 2013 Sep 18. Clin Chim Acta. 2014. PMID: 24055369 Review.
Cited by
-
Peritruncal coronary endothelial cells contribute to proximal coronary artery stems and their aortic orifices in the mouse heart.PLoS One. 2013 Nov 21;8(11):e80857. doi: 10.1371/journal.pone.0080857. eCollection 2013. PLoS One. 2013. PMID: 24278332 Free PMC article.
-
Endothelium as a gatekeeper of fatty acid transport.Trends Endocrinol Metab. 2014 Feb;25(2):99-106. doi: 10.1016/j.tem.2013.11.001. Epub 2013 Dec 3. Trends Endocrinol Metab. 2014. PMID: 24315207 Free PMC article.
-
An endothelial apelin-FGF link mediated by miR-424 and miR-503 is disrupted in pulmonary arterial hypertension.Nat Med. 2013 Jan;19(1):74-82. doi: 10.1038/nm.3040. Epub 2012 Dec 23. Nat Med. 2013. PMID: 23263626 Free PMC article.
-
Endothelial APLNR regulates tissue fatty acid uptake and is essential for apelin's glucose-lowering effects.Sci Transl Med. 2017 Sep 13;9(407):eaad4000. doi: 10.1126/scitranslmed.aad4000. Sci Transl Med. 2017. PMID: 28904225 Free PMC article.
-
[Pyr1]Apelin-13(1-12) Is a Biologically Active ACE2 Metabolite of the Endogenous Cardiovascular Peptide [Pyr1]Apelin-13.Front Neurosci. 2017 Feb 28;11:92. doi: 10.3389/fnins.2017.00092. eCollection 2017. Front Neurosci. 2017. PMID: 28293165 Free PMC article.
References
-
- Ashley E, Chun HJ, Quertermous T. Opposing cardiovascular roles for the angiotensin and apelin signaling pathways. J Mol Cell Cardiol. 2006;41:778–781. - PubMed
-
- Ashley EA, Powers J, Chen M, Kundu R, Finsterbach T, Caffarelli A, Deng A, Eichhorn J, Mahajan R, Agrawal R, Greve J, Robbins R, Patterson AJ, Bernstein D, Quertermous T. The endogenous peptide apelin potently improves cardiac contractility and reduces cardiac loading in vivo. Cardiovasc Res. 2005;65:73–82. - PMC - PubMed
-
- Bayat H, Swaney JS, Ander AN, Dalton N, Kennedy BP, Hammond HK, Roth DM. Progressive heart failure after myocardial infarction in mice. Basic Res Cardiol. 2002;97:206–213. - PubMed
-
- Berry MF, Pirolli TJ, Jayasankar V, Burdick J, Morine KJ, Gardner TJ, Woo YJ. Apelin has in vivo inotropic effects on normal and failing hearts. Circulation. 2004;110:II187–II193. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

