Electron transport chain of Saccharomyces cerevisiae mitochondria is inhibited by H2O2 at succinate-cytochrome c oxidoreductase level without lipid peroxidation involvement
- PMID: 17907001
- PMCID: PMC3030976
- DOI: 10.1080/10715760701635082
Electron transport chain of Saccharomyces cerevisiae mitochondria is inhibited by H2O2 at succinate-cytochrome c oxidoreductase level without lipid peroxidation involvement
Abstract
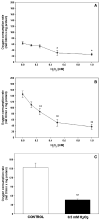
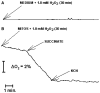
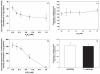
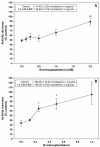
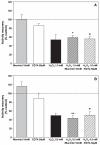
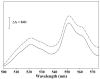
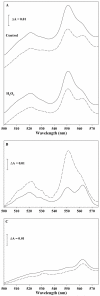
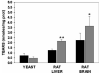
The deleterious effects of H202 on the electron transport chain of yeast mitochondria and on mitochondrial lipid peroxidation were evaluated. Exposure to H2O2 resulted in inhibition of the oxygen consumption in the uncoupled and phosphorylating states to 69% and 65%, respectively. The effect of H2O2 on the respiratory rate was associated with an inhibition of succinate-ubiquinone and succinate-DCIP oxidoreductase activities. Inhibitory effect of H2O2 on respiratory complexes was almost completely recovered by beta-mercaptoethanol treatment. H2O2 treatment resulted in full resistance to Qo site inhibitor myxothiazol and thus it is suggested that the quinol oxidase site (Qo) of complex III is the target for H2O2. H2O2 did not modify basal levels of lipid peroxidation in yeast mitochondria. However, H2O2 addition to rat brain and liver mitochondria induced an increase in lipid peroxidation. These results are discussed in terms of the known physiological differences between mammalian and yeast mitochondria.
Figures
Similar articles
-
Elucidation of the effects of lipoperoxidation on the mitochondrial electron transport chain using yeast mitochondria with manipulated fatty acid content.J Bioenerg Biomembr. 2009 Feb;41(1):15-28. doi: 10.1007/s10863-009-9200-3. Epub 2009 Feb 18. J Bioenerg Biomembr. 2009. PMID: 19224349 Free PMC article.
-
Studies on Hg(II)-induced H2O2 formation and oxidative stress in vivo and in vitro in rat kidney mitochondria.Biochem Pharmacol. 1993 May 25;45(10):2017-24. doi: 10.1016/0006-2952(93)90012-l. Biochem Pharmacol. 1993. PMID: 8512585
-
Extramitochondrial release of hydrogen peroxide from insect and mouse liver mitochondria using the respiratory inhibitors phosphine, myxothiazol, and antimycin and spectral analysis of inhibited cytochromes.Arch Biochem Biophys. 1990 Apr;278(1):65-72. doi: 10.1016/0003-9861(90)90232-n. Arch Biochem Biophys. 1990. PMID: 2321971
-
Mercury-induced H2O2 production and lipid peroxidation in vitro in rat kidney mitochondria.Biochem Pharmacol. 1991 Dec 11;42 Suppl:S181-7. doi: 10.1016/0006-2952(91)90408-w. Biochem Pharmacol. 1991. PMID: 1768276
-
[Superoxide formation and lipid peroxidation by the mitochondrial electron-transfer chain].Rinsho Shinkeigaku. 1994 Dec;34(12):1269-71. Rinsho Shinkeigaku. 1994. PMID: 7774132 Review. Japanese.
Cited by
-
A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis.Proc Natl Acad Sci U S A. 2008 Sep 23;105(38):14447-52. doi: 10.1073/pnas.0803790105. Epub 2008 Sep 15. Proc Natl Acad Sci U S A. 2008. PMID: 18794531 Free PMC article.
-
Elucidation of the effects of lipoperoxidation on the mitochondrial electron transport chain using yeast mitochondria with manipulated fatty acid content.J Bioenerg Biomembr. 2009 Feb;41(1):15-28. doi: 10.1007/s10863-009-9200-3. Epub 2009 Feb 18. J Bioenerg Biomembr. 2009. PMID: 19224349 Free PMC article.
-
Protective effects of resveratrol on calcium-induced oxidative stress in rat heart mitochondria.J Bioenerg Biomembr. 2011 Apr;43(2):101-7. doi: 10.1007/s10863-011-9349-4. Epub 2011 Mar 30. J Bioenerg Biomembr. 2011. PMID: 21448653
-
Phospholipid biosynthesis disruption renders the yeast cells sensitive to antifungals.Folia Microbiol (Praha). 2020 Feb;65(1):121-131. doi: 10.1007/s12223-019-00713-3. Epub 2019 May 15. Folia Microbiol (Praha). 2020. PMID: 31093957
-
Electron transport chain dysfunction by H(2)O (2) is linked to increased reactive oxygen species production and iron mobilization by lipoperoxidation: studies using Saccharomyces cerevisiae mitochondria.J Bioenerg Biomembr. 2011 Apr;43(2):135-47. doi: 10.1007/s10863-011-9339-6. Epub 2011 Feb 25. J Bioenerg Biomembr. 2011. PMID: 21350953
References
-
- Beckman KB, Ames BN. The free radical theory of aging matures. Phys Rev. 1998;78:547–581. - PubMed
-
- Nishikawa T, Edelstein D, Du XL, Yamagishi SI, Matsumura T, Kaneda Y, Yorek MA, Beebe D, Oates PJ, Hammes H-P, Giardino I, Brownlee M. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature. 2000;404:787–790. - PubMed
-
- Starkov AA, Chinopoulos C, Fiskum G. Mitochondrial calcium and oxidative stress as mediators of ischemic brain injury. Cell Calcium. 2004;36:257–264. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
