Apoptotic killing of HIV-1-infected macrophages is subverted by the viral envelope glycoprotein
- PMID: 17907802
- PMCID: PMC2323301
- DOI: 10.1371/journal.ppat.0030134
Apoptotic killing of HIV-1-infected macrophages is subverted by the viral envelope glycoprotein
Abstract
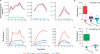
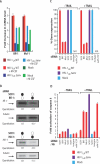
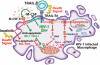
Viruses have evolved strategies to protect infected cells from apoptotic clearance. We present evidence that HIV-1 possesses a mechanism to protect infected macrophages from the apoptotic effects of the death ligand TRAIL (tumor necrosis factor-related apoptosis-inducing ligand). In HIV-1-infected macrophages, the viral envelope protein induced macrophage colony-stimulating factor (M-CSF). This pro-survival cytokine downregulated the TRAIL receptor TRAIL-R1/DR4 and upregulated the anti-apoptotic genes Bfl-1 and Mcl-1. Inhibition of M-CSF activity or silencing of Bfl-1 and Mcl-1 rendered infected macrophages highly susceptible to TRAIL. The anti-cancer agent Imatinib inhibited M-CSF receptor activation and restored the apoptotic sensitivity of HIV-1-infected macrophages, suggesting a novel strategy to curtail viral persistence in the macrophage reservoir.
Conflict of interest statement
Figures




References
-
- Ray N, Doms RW. HIV-1 coreceptors and their inhibitors. Curr Top Microbiol Immunol. 2006;303:97–120. - PubMed
-
- Cicala C, Arthos J, Ruiz M, Vaccarezza M, Rubbert A, et al. Induction of phosphorylation and intracellular association of CC chemokine receptor 5 and focal adhesion kinase in primary human CD4+ T cells by macrophage-tropic HIV envelope. J Immunol. 1999;163:420–426. - PubMed
-
- Kaplan MJ, Ray D, Mo RR, Yung RL, Richardson BC. TRAIL (Apo2 ligand) and TWEAK (Apo3 ligand) mediate CD4+ T cell killing of antigen-presenting macrophages. J Immunol. 2000;164:2897–2904. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

