Kaposi's sarcoma herpesvirus-encoded latency-associated nuclear antigen stabilizes intracellular activated Notch by targeting the Sel10 protein
- PMID: 17909182
- PMCID: PMC1997218
- DOI: 10.1073/pnas.0703508104
Kaposi's sarcoma herpesvirus-encoded latency-associated nuclear antigen stabilizes intracellular activated Notch by targeting the Sel10 protein
Abstract
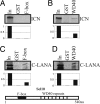
Deregulation of the evolutionarily conserved Notch signaling is highly correlated with oncogenesis. Intracellular activated Notch (ICN) is a protooncogene linked to the transcription activation of a number of cellular genes involved in cell cycle regulation, differentiation, and proliferation. Stability of ICN is tightly regulated by the Sel10-mediated ubiquitin-proteasome pathway. Sel10 can function as a negative regulator of Notch and exhibits activities of a tumor-suppressor protein. This article shows that the Kaposi's sarcoma-associated herpesvirus (KSHV) latency-associated nuclear antigen (LANA) directly interacts with Sel10 and forms a complex in KSHV-infected cells. This results in suppression of ICN ubiquitination and degradation. The carboxyl terminus of LANA interacts with the F-box and WD40 domains of Sel10 and competes with ICN for binding to Sel10. This elevated level of ICN is also critical for maintaining the enhanced proliferation of KSHV-infected tumor cells. These findings describe a mechanism by which the KSHV-encoded LANA protein regulates ubiquitination of ICN mediated by the F-box component of the E3 ligase Sel10, leading to proliferation of the virus-infected cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

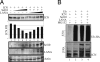

Similar articles
-
Kaposi's Sarcoma-Associated Herpesvirus LANA Modulates the Stability of the E3 Ubiquitin Ligase RLIM.J Virol. 2020 Feb 14;94(5):e01578-19. doi: 10.1128/JVI.01578-19. Print 2020 Feb 14. J Virol. 2020. PMID: 31801865 Free PMC article.
-
Bub1 in Complex with LANA Recruits PCNA To Regulate Kaposi's Sarcoma-Associated Herpesvirus Latent Replication and DNA Translesion Synthesis.J Virol. 2015 Oct;89(20):10206-18. doi: 10.1128/JVI.01524-15. Epub 2015 Jul 29. J Virol. 2015. PMID: 26223641 Free PMC article.
-
Protein complexes associated with the Kaposi's sarcoma-associated herpesvirus-encoded LANA.Virology. 2007 Aug 1;364(2):317-29. doi: 10.1016/j.virol.2007.03.010. Epub 2007 Apr 16. Virology. 2007. PMID: 17434559 Free PMC article.
-
Regulation of Notch signaling by E3 ubiquitin ligases.FEBS J. 2022 Feb;289(4):937-954. doi: 10.1111/febs.15792. Epub 2021 Mar 16. FEBS J. 2022. PMID: 33644958 Review.
-
Natural killer cell evasion by an E3 ubiquitin ligase from Kaposi's sarcoma-associated herpesvirus.Biochem Soc Trans. 2008 Jun;36(Pt 3):459-63. doi: 10.1042/BST0360459. Biochem Soc Trans. 2008. PMID: 18481981 Review.
Cited by
-
The latency-associated nuclear antigen, a multifunctional protein central to Kaposi's sarcoma-associated herpesvirus latency.Future Microbiol. 2011 Dec;6(12):1399-413. doi: 10.2217/fmb.11.137. Future Microbiol. 2011. PMID: 22122438 Free PMC article. Review.
-
FBXW7: a critical tumor suppressor of human cancers.Mol Cancer. 2018 Aug 7;17(1):115. doi: 10.1186/s12943-018-0857-2. Mol Cancer. 2018. PMID: 30086763 Free PMC article. Review.
-
E3 Ubiquitin Ligases in Gammaherpesviruses and HIV: A Review of Virus Adaptation and Exploitation.Viruses. 2023 Sep 15;15(9):1935. doi: 10.3390/v15091935. Viruses. 2023. PMID: 37766341 Free PMC article. Review.
-
Opposing regulation of PROX1 by interleukin-3 receptor and NOTCH directs differential host cell fate reprogramming by Kaposi sarcoma herpes virus.PLoS Pathog. 2012;8(6):e1002770. doi: 10.1371/journal.ppat.1002770. Epub 2012 Jun 14. PLoS Pathog. 2012. PMID: 22719258 Free PMC article.
-
Onco-Pathogen Mediated Cancer Progression and Associated Signaling Pathways in Cancer Development.Pathogens. 2023 May 28;12(6):770. doi: 10.3390/pathogens12060770. Pathogens. 2023. PMID: 37375460 Free PMC article. Review.
References
-
- Chang Y, Cesarman E, Pessin MS, Lee F, Culpepper J, Knowles DM, Moore PS. Science. 1994;266:1865–1869. - PubMed
-
- Boshoff C, Chang Y. Annu Rev Med. 2001;52:453–470. - PubMed
-
- Brander C, Raje N, O'Connor PG, Davies F, Davis J, Chauhan D, Hideshima T, Martin J, Osmond D, Kedes DH, et al. Blood. 2002;100:698–700. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

