Targeted deletion of regions rich in immune-evasive genes from the cytomegalovirus genome as a novel vaccine strategy
- PMID: 17913824
- PMCID: PMC2168857
- DOI: 10.1128/JVI.01911-07
Targeted deletion of regions rich in immune-evasive genes from the cytomegalovirus genome as a novel vaccine strategy
Abstract
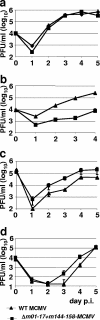
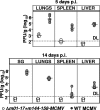
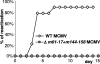
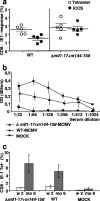
Human cytomegalovirus (CMV), a ubiquitous human pathogen, is a leading cause of congenital infections and represents a serious health risk for the immunosuppressed patient. A vaccine against CMV is currently not available. CMV is characterized by its large genome and by multiple genes modulating the immunity of the host, which cluster predominantly at genome termini. Here, we tested whether the deletion of gene blocks rich in immunomodulatory genes could be used as a novel concept in the generation of immunogenic but avirulent, herpesvirus vaccines. To generate an experimental CMV vaccine, we selectively deleted 32 genes from the mouse cytomegalovirus (MCMV) genome. The resulting mutant grew to titers similar to that of wild-type MCMV in vitro. In vivo, the mutant was 10,000-fold attenuated and well tolerated, even by highly susceptible mice deficient for B, T, and NK cells or for the interferon type I receptor. Equally relevant for safety concerns, immune suppression did not lead to the mutant's reactivation from latency. Immunization with the replication-competent mutant, but not with inactivated virus, resulted in protective immunity, which increased over time. Vaccination induced MCMV-specific antibodies and a strong T-cell response. We propose that a targeted and rational approach can improve future herpesvirus vaccines and vaccine vectors.
Figures




Similar articles
-
Mucosal and parenteral vaccination against acute and latent murine cytomegalovirus (MCMV) infection by using an attenuated MCMV mutant.J Virol. 1998 Jan;72(1):442-51. doi: 10.1128/JVI.72.1.442-451.1998. J Virol. 1998. PMID: 9420244 Free PMC article.
-
Development of a vaccine against murine cytomegalovirus (MCMV), consisting of plasmid DNA and formalin-inactivated MCMV, that provides long-term, complete protection against viral replication.J Virol. 2002 May;76(10):4822-35. doi: 10.1128/jvi.76.10.4822-4835.2002. J Virol. 2002. PMID: 11967299 Free PMC article.
-
Systemic priming-boosting immunization with a trivalent plasmid DNA and inactivated murine cytomegalovirus (MCMV) vaccine provides long-term protection against viral replication following systemic or mucosal MCMV challenge.J Virol. 2005 Jan;79(1):159-75. doi: 10.1128/JVI.79.1.159-175.2005. J Virol. 2005. PMID: 15596812 Free PMC article.
-
Engineering of cytomegalovirus genomes for recombinant live herpesvirus vaccines.Int J Med Microbiol. 2008 Jan;298(1-2):115-25. doi: 10.1016/j.ijmm.2007.07.008. Epub 2007 Aug 16. Int J Med Microbiol. 2008. PMID: 17702650 Review.
-
Conditional gene expression systems to study herpesvirus biology in vivo.Med Microbiol Immunol. 2008 Jun;197(2):269-76. doi: 10.1007/s00430-008-0086-1. Epub 2008 Mar 7. Med Microbiol Immunol. 2008. PMID: 18324415 Review.
Cited by
-
Concatemeric Broccoli reduces mRNA stability and induces aggregates.PLoS One. 2021 Aug 4;16(8):e0244166. doi: 10.1371/journal.pone.0244166. eCollection 2021. PLoS One. 2021. PMID: 34347781 Free PMC article.
-
A spread-deficient cytomegalovirus for assessment of first-target cells in vaccination.J Virol. 2010 Aug;84(15):7730-42. doi: 10.1128/JVI.02696-09. Epub 2010 May 12. J Virol. 2010. PMID: 20463067 Free PMC article.
-
Can we build it better? Using BAC genetics to engineer more effective cytomegalovirus vaccines.J Clin Invest. 2010 Dec;120(12):4192-7. doi: 10.1172/JCI45250. Epub 2010 Nov 22. J Clin Invest. 2010. PMID: 21099107 Free PMC article.
-
MCMV avoidance of recognition and control by NK cells.Semin Immunopathol. 2014 Nov;36(6):641-50. doi: 10.1007/s00281-014-0441-9. Epub 2014 Aug 21. Semin Immunopathol. 2014. PMID: 25141793 Review.
-
Developing a Vaccine against Congenital Cytomegalovirus (CMV) Infection: What Have We Learned from Animal Models? Where Should We Go Next?Future Virol. 2013 Dec;8(12):1161-1182. doi: 10.2217/fvl.13.106. Future Virol. 2013. PMID: 24523827 Free PMC article.
References
-
- Adler, S. P., S. H. Hempfling, S. E. Starr, S. A. Plotkin, and S. Riddell. 1998. Safety and immunogenicity of the Towne strain cytomegalovirus vaccine. Pediatr. Infect. Dis. J. 17:200-206. - PubMed
-
- Adler, S. P., S. E. Starr, S. A. Plotkin, S. H. Hempfling, J. Buis, M. L. Manning, and A. M. Best. 1995. Immunity induced by primary human cytomegalovirus infection protects against secondary infection among women of childbearing age. J. Infect. Dis. 171:26-32. - PubMed
-
- Arase, H., E. S. Mocarski, A. E. Campbell, A. B. Hill, and L. L. Lanier. 2002. Direct recognition of cytomegalovirus by activating and inhibitory NK cell receptors. Science 296:1323-1326. - PubMed
-
- Boppana, S. B., L. B. Rivera, K. B. Fowler, M. Mach, and W. J. Britt. 2001. Intrauterine transmission of cytomegalovirus to infants of women with preconceptional immunity. N. Engl. J. Med. 344:1366-1371. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

