AP-1 and ARF1 control endosomal dynamics at sites of FcR mediated phagocytosis
- PMID: 17914058
- PMCID: PMC2096587
- DOI: 10.1091/mbc.e07-04-0392
AP-1 and ARF1 control endosomal dynamics at sites of FcR mediated phagocytosis
Abstract
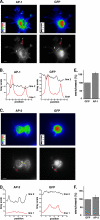
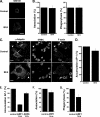
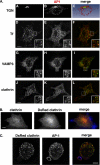
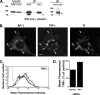
Phagocytosis, the mechanism of ingestion of large material and microorganisms, relies on actin polymerization and on the focal delivery of intracellular endocytic compartments. The molecular mechanisms involved in the formation and delivery of the endocytic vesicles that are recruited at sites of phagocytosis are not well characterized. Here we show that adaptor protein (AP)-1 but not AP-2 clathrin adaptor complexes are recruited early below the sites of particle attachment and are required for efficient receptor-mediated phagocytosis in murine macrophages. Clathrin, however, is not recruited with the AP complexes. We further show that the recruitment of AP-1-positive structures at sites of phagocytosis is regulated by the GTP-binding protein ARF1 but is not sensitive to brefeldin A. Furthermore, AP-1 depletion leads to increased surface levels of TNF-alpha, a cargo known to traffic through the endosomes to the plasma membrane upon stimulation of the macrophages. Together, our results support a clathrin-independent role for AP complexes in endosomal dynamics in macrophages by retaining some cargo proteins, a process important for membrane remodeling during phagocytosis.
Figures


Similar articles
-
Binding of cargo sorting signals to AP-1 enhances its association with ADP ribosylation factor 1-GTP.J Cell Biol. 2008 Feb 11;180(3):467-72. doi: 10.1083/jcb.200709037. Epub 2008 Feb 4. J Cell Biol. 2008. PMID: 18250197 Free PMC article.
-
Structural basis for recruitment and activation of the AP-1 clathrin adaptor complex by Arf1.Cell. 2013 Feb 14;152(4):755-67. doi: 10.1016/j.cell.2012.12.042. Cell. 2013. PMID: 23415225 Free PMC article.
-
In vivo monitoring of the recruitment and activation of AP-1 by Arf1.Sci Rep. 2017 Aug 2;7(1):7148. doi: 10.1038/s41598-017-07493-1. Sci Rep. 2017. PMID: 28769048 Free PMC article.
-
The clathrin adaptor complexes as a paradigm for membrane-associated allostery.Protein Sci. 2013 May;22(5):517-29. doi: 10.1002/pro.2235. Epub 2013 Mar 18. Protein Sci. 2013. PMID: 23424177 Free PMC article. Review.
-
Cargo adaptors: structures illuminate mechanisms regulating vesicle biogenesis.Trends Cell Biol. 2015 Jul;25(7):408-16. doi: 10.1016/j.tcb.2015.02.005. Epub 2015 Mar 17. Trends Cell Biol. 2015. PMID: 25795254 Free PMC article. Review.
Cited by
-
Comparative Anatomy of Phagocytic and Immunological Synapses.Front Immunol. 2016 Jan 28;7:18. doi: 10.3389/fimmu.2016.00018. eCollection 2016. Front Immunol. 2016. PMID: 26858721 Free PMC article. Review.
-
Phosphoinositide 3-kinase δ regulates membrane fission of Golgi carriers for selective cytokine secretion.J Cell Biol. 2010 Sep 20;190(6):1053-65. doi: 10.1083/jcb.201001028. Epub 2010 Sep 13. J Cell Biol. 2010. PMID: 20837769 Free PMC article.
-
Lysoplex: An efficient toolkit to detect DNA sequence variations in the autophagy-lysosomal pathway.Autophagy. 2015;11(6):928-38. doi: 10.1080/15548627.2015.1043077. Autophagy. 2015. PMID: 26075876 Free PMC article.
-
Phagocytosis at a glance.J Cell Sci. 2025 Jun 15;138(12):jcs263833. doi: 10.1242/jcs.263833. Epub 2025 Jul 1. J Cell Sci. 2025. PMID: 40590685 Free PMC article. Review.
-
Origin of the cell nucleus, mitosis and sex: roles of intracellular coevolution.Biol Direct. 2010 Feb 4;5:7. doi: 10.1186/1745-6150-5-7. Biol Direct. 2010. PMID: 20132544 Free PMC article.
References
-
- Aderem A. How to eat something bigger than your head. Cell. 2002;110:5–8. - PubMed
-
- Aderem A., Underhill D. M. Mechanisms of phagocytosis in macrophages. Annu. Rev. Immunol. 1999;17:593–623. - PubMed
-
- Araki N., Hatae T., Furukawa A., Swanson J. A. Phosphoinositide-3-kinase-independent contractile activities associated with Fcgamma-receptor-mediated phagocytosis and macropinocytosis in macrophages. J. Cell Sci. 2003;116:247–257. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

