Peptide-functionalized poly(ethylene glycol) star polymers: DNA delivery vehicles with multivalent molecular architecture
- PMID: 17915935
- PMCID: PMC2650482
- DOI: 10.1021/bc0701141
Peptide-functionalized poly(ethylene glycol) star polymers: DNA delivery vehicles with multivalent molecular architecture
Abstract
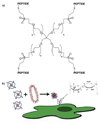
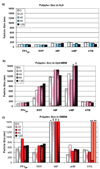
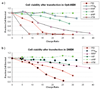
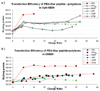
Exploring the development of nonviral nucleic acid delivery vectors with progressive, specific, and novel designs in molecular architecture is a fundamental way to investigate how aspects of chemical and physical structure impact the transfection process. In this study, macromolecules comprised of a four-arm star poly(ethylene glycol) and termini modified with one of five different heparin binding peptides have been investigated for their ability to bind, compact, and deliver DNA to mammalian cells in vitro. These new delivery vectors combine a PEG-derived stabilizing moiety with peptides that exhibit unique cell-surface binding ability in a molecular architecture that permits multivalent presentation of the cationic peptides. Five peptide sequences of varying heparin binding affinity were studied; each was found to sufficiently bind heparin for biological application. Additionally, the macromolecules were able to bind and compact DNA into particles of proper size for endocytosis. In biological studies, the PEG-star peptides displayed a range of toxicity and transfection efficiency dependent on the peptide identity. The vectors equipped with peptides of highest heparin binding affinity were found to bind DNA tightly, increase levels of cellular internalization, and display the most promising transfection qualities. Our results suggest heparin binding peptides with specific sequences hold more potential than nonspecific cationic polymers to optimize transfection efficiency while maintaining cell viability. Furthermore, the built-in multivalency of these macromolecules may allow simultaneous binding of both DNA at the core of the polyplex and heparan sulfate on the surface of the cell. This scheme may facilitate a bridging transport mechanism, tethering DNA to the surface of the cell and subsequently ushering therapeutic nucleic acids into the cell. This multivalent star shape is therefore a promising architectural feature that may be exploited in the design of future polycationic gene delivery vectors.
Figures


Similar articles
-
Synthesis and characterization of four-arm poly(ethylene glycol)-based gene delivery vehicles coupled to integrin and DNA-binding peptides.Mol Pharm. 2008 Jan-Feb;5(1):140-50. doi: 10.1021/mp700072n. Epub 2007 Dec 13. Mol Pharm. 2008. PMID: 18076138
-
Characterization of a multifunctional PEG-based gene delivery system containing nuclear localization signals and endosomal escape peptides.Acta Biomater. 2009 Mar;5(3):854-64. doi: 10.1016/j.actbio.2008.09.009. Epub 2008 Sep 30. Acta Biomater. 2009. PMID: 18926782
-
Folic Acid/Peptides Modified PLGA-PEI-PEG Polymeric Vectors as Efficient Gene Delivery Vehicles: Synthesis, Characterization and Their Biological Performance.Mol Biotechnol. 2021 Jan;63(1):63-79. doi: 10.1007/s12033-020-00285-5. Epub 2020 Nov 3. Mol Biotechnol. 2021. PMID: 33141343
-
Oligoarginine-PEG-lipid particles for gene delivery.Expert Opin Drug Deliv. 2009 Oct;6(10):1065-77. doi: 10.1517/17425240903156366. Expert Opin Drug Deliv. 2009. PMID: 19637981 Review.
-
Internalization of cationic peptides: the road less (or more?) traveled.Cell Mol Life Sci. 2006 Aug;63(16):1819-22. doi: 10.1007/s00018-006-6170-z. Cell Mol Life Sci. 2006. PMID: 16909213 Free PMC article. Review.
Cited by
-
Star-polymer unimolecular micelle nanoparticles to deliver a payload across the blood-brain barrier.Nanoscale. 2024 Nov 28;16(46):21582-21593. doi: 10.1039/d4nr02636e. Nanoscale. 2024. PMID: 39494464 Free PMC article.
-
Polymeric nucleic acid vehicles exploit active interorganelle trafficking mechanisms.ACS Nano. 2013 Jan 22;7(1):347-64. doi: 10.1021/nn304218q. Epub 2012 Dec 31. ACS Nano. 2013. PMID: 23234474 Free PMC article.
-
Polymer-Based Therapeutics.Macromolecules. 2009 Jan 13;42(1):3-13. doi: 10.1021/ma801782q. Macromolecules. 2009. PMID: 21494423 Free PMC article.
-
Structurally nanoengineered antimicrobial peptide polymers: design, synthesis and biomedical applications.World J Microbiol Biotechnol. 2021 Jul 19;37(8):139. doi: 10.1007/s11274-021-03109-z. World J Microbiol Biotechnol. 2021. PMID: 34278535 Free PMC article. Review.
-
Applications of orthogonal "click" chemistries in the synthesis of functional soft materials.Chem Rev. 2009 Nov;109(11):5620-86. doi: 10.1021/cr900138t. Chem Rev. 2009. PMID: 19905010 Free PMC article. Review. No abstract available.
References
-
- Akhtar S. Non-viral cancer gene therapy: Beyond delivery. Gene Ther. 2006;13:739–740. - PubMed
-
- Reineke TM, Grinstaff MW. Designer materials for nucleic acid delivery. MRS Bull. 2005;30:635–638.
-
- Deshpande MC, Davies MC, Garnett MC, Williams PM, Armitage D, Bailey L, Vamvakaki M, Armes SP, Stolnik S. The effect of poly(ethylene glycol molecular architecture on cellular interaction and uptake of DNA complexes. J. Controlled Release. 2004;97:143–156. - PubMed
-
- Liu Y, Wenning L, Lynch M, Reineke TM. New poly(D-glucaramidoamine)s induce DNA nanoparticle formation and efficient gene delivery into mammalian cells. J. Am. Chem. Soc. 2004;126:7422–7423. - PubMed
-
- Liu YM, Reineke TM. Hydroxyl stereochemistry and amine number within poly(glycoamidoamine)s affect intracellular DNA delivery. J. Am. Chem. Soc. 2005;127:3004–3015. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

