Neither DNA hypomethylation nor changes in the kinetics of erythroid differentiation explain 5-azacytidine's ability to induce human fetal hemoglobin
- PMID: 17916742
- PMCID: PMC2200821
- DOI: 10.1182/blood-2007-06-093948
Neither DNA hypomethylation nor changes in the kinetics of erythroid differentiation explain 5-azacytidine's ability to induce human fetal hemoglobin
Abstract
5-azacytidine (5-Aza) is a potent inducer of fetal hemoglobin (HbF) in people with beta-thalassemia and sickle cell disease. Two models have been proposed to explain this activity. The first is based on the drug's ability to inhibit global DNA methylation, including the fetal globin genes, resulting in their activation. The second is based on 5-Aza's cytotoxicity and observations that HbF production is enhanced during marrow recovery. We tested these models using human primary cells in an in vitro erythroid differentiation system. We found that doses of 5-Aza that produce near maximal induction of gamma-globin mRNA and HbF do not alter cell growth, differentiation kinetics, or cell cycle, but do cause a localized demethylation of the gamma promoter. However, when we reduced gamma promoter methylation to levels equivalent to those seen with 5-Aza or to the lower levels seen in primary fetal erythroid cells using DNMT1 siRNA and shRNA, we observed no induction of gamma-globin mRNA or HbF. These results suggest that 5-Aza induction of HbF is not the result of global DNA demethylation or of changes in differentiation kinetics, but involves an alternative, previously unrecognized mechanism. Other results suggest that posttranscriptional regulation plays an important role in the 5-Aza response.
Figures


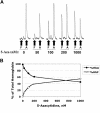

 ) and without (□) 300 nM 5-azacytidine treatment. For comparison, the methylation of the CpGs at day +5 without 5-azacytidine is shown (▨). (C) Methylation of individual γ-globin CpGs on day +14. (D) Methylation of individual γ-globin CpGs on day +18. (D) Comparison of total γ-globin promoter methylation during differentiation with and without 300 nM 5-azacytidine. (E) Comparison of total γ-globin promoter methylation during differentiation with and without 300 nM 5-azacytidine. (F) As a measure of genome-wide DNA methylation, the effect of 300 nM 5-azacytidine on LINE methylation was determined. CD34+ cells from a single normal donor were used for this experiment. Error bars represent 1 SD. P values were determined by t test.
) and without (□) 300 nM 5-azacytidine treatment. For comparison, the methylation of the CpGs at day +5 without 5-azacytidine is shown (▨). (C) Methylation of individual γ-globin CpGs on day +14. (D) Methylation of individual γ-globin CpGs on day +18. (D) Comparison of total γ-globin promoter methylation during differentiation with and without 300 nM 5-azacytidine. (E) Comparison of total γ-globin promoter methylation during differentiation with and without 300 nM 5-azacytidine. (F) As a measure of genome-wide DNA methylation, the effect of 300 nM 5-azacytidine on LINE methylation was determined. CD34+ cells from a single normal donor were used for this experiment. Error bars represent 1 SD. P values were determined by t test.


Comment in
-
DNA methylation and mechanism of action of 5-azacytidine.Blood. 2008 Feb 15;111(4):2485; author reply 2486. doi: 10.1182/blood-2007-10-119867. Blood. 2008. PMID: 18263785 No abstract available.
Similar articles
-
Hydroxymethylcytosine and demethylation of the γ-globin gene promoter during erythroid differentiation.Epigenetics. 2015;10(5):397-407. doi: 10.1080/15592294.2015.1039220. Epigenetics. 2015. PMID: 25932923 Free PMC article.
-
Plastrum testudinis induces γ-globin gene expression through epigenetic histone modifications within the γ-globin gene promoter via activation of the p38 MAPK signaling pathway.Int J Mol Med. 2013 Jun;31(6):1418-28. doi: 10.3892/ijmm.2013.1338. Epub 2013 Apr 8. Int J Mol Med. 2013. PMID: 23588991
-
Metformin induces FOXO3-dependent fetal hemoglobin production in human primary erythroid cells.Blood. 2018 Jul 19;132(3):321-333. doi: 10.1182/blood-2017-11-814335. Epub 2018 Jun 8. Blood. 2018. PMID: 29884740 Free PMC article.
-
Clinical studies with fetal hemoglobin-enhancing agents in sickle cell disease.Semin Hematol. 2004 Oct;41(4 Suppl 6):11-6. doi: 10.1053/j.seminhematol.2004.08.003. Semin Hematol. 2004. PMID: 15534852 Review.
-
Induction of hemoglobin F synthesis in patients with beta thalassemia.Annu Rev Med. 1985;36:485-98. doi: 10.1146/annurev.me.36.020185.002413. Annu Rev Med. 1985. PMID: 2581500 Review.
Cited by
-
Compound loss of function of nuclear receptors Tr2 and Tr4 leads to induction of murine embryonic β-type globin genes.Blood. 2015 Feb 26;125(9):1477-87. doi: 10.1182/blood-2014-10-605022. Epub 2015 Jan 5. Blood. 2015. PMID: 25561507 Free PMC article.
-
Revisiting fetal hemoglobin inducers in beta-hemoglobinopathies: a review of natural products, conventional and combinatorial therapies.Mol Biol Rep. 2022 Mar;49(3):2359-2373. doi: 10.1007/s11033-021-06977-8. Epub 2021 Nov 25. Mol Biol Rep. 2022. PMID: 34822068 Review.
-
Induction of human fetal hemoglobin via the NRF2 antioxidant response signaling pathway.Blood. 2011 Jun 2;117(22):5987-97. doi: 10.1182/blood-2010-10-314096. Epub 2011 Apr 4. Blood. 2011. PMID: 21464371 Free PMC article.
-
S110, a novel decitabine dinucleotide, increases fetal hemoglobin levels in baboons (P. anubis).J Transl Med. 2010 Oct 8;8:92. doi: 10.1186/1479-5876-8-92. J Transl Med. 2010. PMID: 20932295 Free PMC article.
-
Developmentally regulated extended domains of DNA hypomethylation encompass highly transcribed genes of the human beta-globin locus.Exp Hematol. 2009 Jul;37(7):807-813.e2. doi: 10.1016/j.exphem.2009.04.005. Epub 2009 May 19. Exp Hematol. 2009. PMID: 19460471 Free PMC article.
References
-
- Ali SA. Milder variant of sickle-cell disease in Arabs in Kuwait associated with unusually high level of foetal haemoglobin. Br J Haematol. 1970;19:613–619. - PubMed
-
- Prchal J, Stamatoyannopoulos G. Two siblings with unusually mild homozygous beta-thalassemia: a didactic example of the effect of a nonallelic modifier gene of the expressivity of a monogenic disorder. Am J Med Genet. 1981;10:291–300. - PubMed
-
- Pace BS, Zein S. Understanding mechanisms of gamma-globin gene regulation to develop strategies for pharmacological fetal hemoglobin induction. Dev Dyn. 2006;235:1727–1737. - PubMed
-
- Ley TJ, DeSimone J, Anagnou NP, et al. 5-azacytidine selectively increases gamma-globin synthesis in a patient with beta+ thalassemia. N Engl J Med. 1982;307:1469–1475. - PubMed
-
- Dover GJ, Charache SH, Boyer SH, Talbot CC, Jr., Smith KD. 5-Azacytidine increases fetal hemoglobin production in a patient with sickle cell disease. Prog Clin Biol Res. 1983;134:475–488. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

