Reciprocal effects of micro-RNA-122 on expression of heme oxygenase-1 and hepatitis C virus genes in human hepatocytes
- PMID: 17919492
- PMCID: PMC2128739
- DOI: 10.1053/j.gastro.2007.08.002
Reciprocal effects of micro-RNA-122 on expression of heme oxygenase-1 and hepatitis C virus genes in human hepatocytes
Abstract
Background & aims: Heme oxygenase-1 (HO-1) is an antioxidant defense and key cytoprotective enzyme, which is repressed by Bach1. Micro-RNA-122 (miR-122) is specifically expressed and highly abundant in human liver and required for replication of hepatitis C virus (HCV) RNA. This study was to assess whether a specific miR-122 antagomir down-regulates HCV protein replication and up-regulates HO-1.
Methods: We transfected antagomir of miR-122, 2'-O-methyl-mimic miR-122, or nonspecific control antagomir, into wild-type (WT) Huh-7 cells or Huh-7 stably replicating HCV subgenomic protein core through nonstructural protein 3 of HCV (NS3) (CNS3 replicon cells) or NS3-5B (9-13 replicon cells).
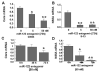
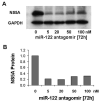
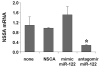
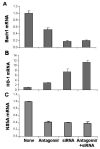
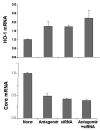
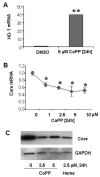
Results: Antagomir of miR-122 reduced the abundance of HCV RNA by 64% in CNS3 and by 84% in 9-13 cells. Transfection with 2'-O-methlyl-mimic miR-122 increased HCV levels up to 2.5-fold. Antagomir of miR-122 also decreased Bach1 and increased HO-1 mRNA levels in CNS3, 9-13, and WT Huh-7 cells. Increasing HO-1 by silencing Bach1 with 50 nmol/L Bach1-short interfering RNA or by treatment with 5 mumol/L cobalt protoporphyrin or heme (known inducers of HO-1) decreased HCV RNA and protein by 50% in HCV replicon cells.
Conclusions: Down-regulation of HCV replication using an antagomir targeted to miR-122 is effective, specific, and selective. Increasing HO-1, by silencing the Bach1 gene or by treatment with cobalt protoporphyrin or heme, decreases HCV replication. Thus, miR-122 plays an important role in the regulation of HCV replication and HO-1/Bach1 expression in hepatocytes. Down-regulation of miR-122 and up-regulation of HO-1 may be new strategies for anti-HCV intervention and cytoprotection.
Conflict of interest statement
No conflict of interest exist.
Figures

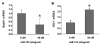
Similar articles
-
MicroRNA-196 represses Bach1 protein and hepatitis C virus gene expression in human hepatoma cells expressing hepatitis C viral proteins.Hepatology. 2010 May;51(5):1494-504. doi: 10.1002/hep.23401. Hepatology. 2010. PMID: 20127796 Free PMC article.
-
HCV proteins increase expression of heme oxygenase-1 (HO-1) and decrease expression of Bach1 in human hepatoma cells.J Hepatol. 2006 Jul;45(1):5-12. doi: 10.1016/j.jhep.2005.12.020. Epub 2006 Feb 3. J Hepatol. 2006. PMID: 16530877
-
Role of Bach1 and Nrf2 in up-regulation of the heme oxygenase-1 gene by cobalt protoporphyrin.FASEB J. 2006 Dec;20(14):2651-3. doi: 10.1096/fj.06-6346fje. Epub 2006 Oct 25. FASEB J. 2006. PMID: 17065227
-
Legalon-SIL downregulates HCV core and NS5A in human hepatocytes expressing full-length HCV.World J Gastroenterol. 2011 Apr 7;17(13):1694-700. doi: 10.3748/wjg.v17.i13.1694. World J Gastroenterol. 2011. PMID: 21483629 Free PMC article.
-
New biotechnological methods to reduce oxidative stress in the cardiovascular system: focusing on the Bach1/heme oxygenase-1 pathway.Curr Pharm Biotechnol. 2006 Apr;7(2):87-93. doi: 10.2174/138920106776597630. Curr Pharm Biotechnol. 2006. PMID: 16724942 Review.
Cited by
-
Oxidative stress response and Nrf2 signaling in aging.Free Radic Biol Med. 2015 Nov;88(Pt B):314-336. doi: 10.1016/j.freeradbiomed.2015.05.036. Epub 2015 Jun 9. Free Radic Biol Med. 2015. PMID: 26066302 Free PMC article. Review.
-
Micro-Players of Great Significance-Host microRNA Signature in Viral Infections in Humans and Animals.Int J Mol Sci. 2022 Sep 11;23(18):10536. doi: 10.3390/ijms231810536. Int J Mol Sci. 2022. PMID: 36142450 Free PMC article. Review.
-
Heme oxygenase-1 as a therapeutic target in inflammatory disorders of the gastrointestinal tract.World J Gastroenterol. 2010 Jul 7;16(25):3112-9. doi: 10.3748/wjg.v16.i25.3112. World J Gastroenterol. 2010. PMID: 20593496 Free PMC article. Review.
-
The roles of different microRNAs in the regulation of cholesterol in viral hepatitis.Cell Commun Signal. 2023 Sep 14;21(1):231. doi: 10.1186/s12964-023-01250-w. Cell Commun Signal. 2023. PMID: 37710249 Free PMC article. Review.
-
Regulation of heme oxygenase-1 protein expression by miR-377 in combination with miR-217.J Biol Chem. 2011 Feb 4;286(5):3194-202. doi: 10.1074/jbc.M110.148726. Epub 2010 Nov 24. J Biol Chem. 2011. PMID: 21106538 Free PMC article.
References
-
- Shepard CW, Finelli L, Alter M. Global epidemiology of hepatitis C virus infection. Lancet Infectious Diseases. 2005;5:558–567. - PubMed
-
- Shiffman ML, Saab S, Feng S, Abecassis MI, Tzakis AG, Goodrich NP, Schaubel DE. Liver and intestine transplantation in the United States, 1995–2004. Am J Transplant. 2006;6:1170–1187. - PubMed
-
- Lieber CS. Role of oxidative stress and antioxidant therapy in alcoholic and nonalcoholic liver diseases. Adv Pharmacol. 1997;38:601–628. - PubMed
-
- Tsutsumi T, Suzuki T, Moriya K, Shintani Y, Fujie H, Miyoshi H, Matsuura Y, Koike K, Miyamura T. Hepatitis C virus core protein activates ERK and p38 MAPK in cooperation with ethanol in transgenic mice. Hepatology. 2003;38:820–828. - PubMed
-
- Wen F, Abdalla MY, Aloman C, Xiang J, Ahmad IM, Walewski J, McCormick ML, Brown KE, Branch AD, Spitz DR, Britigan BE, Schmidt WN. Increased prooxidant production and enhanced susceptibility to glutathione depletion in HepG2 cells co-expressing HCV core protein and CYP2E1. J Med Virol. 2004;72:230–240. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

