The ferric-hydroperoxo complex of chloroperoxidase
- PMID: 17920039
- PMCID: PMC2078203
- DOI: 10.1016/j.bbrc.2007.09.085
The ferric-hydroperoxo complex of chloroperoxidase
Abstract
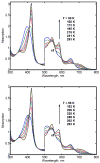
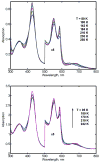
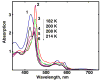
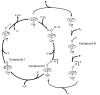
The hydroperoxo-ferric complex, or Compound 0 (Cpd 0), is an unstable transient intermediate common for oxygen activating heme enzymes such as the cytochromes P450, nitric oxide synthases, and heme oxygenases, as well as the peroxidases and catalases which utilize hydrogen peroxide as a source of oxygen and reducing equivalents. Detailed understanding of the mechanism of oxygen activation and formation of the higher valent catalytically active intermediates in heme enzyme catalysis requires the structural and spectroscopic characterization of this immediate precursor, Cpd 0. Using the method of cryoradiolytic reduction of the oxy-ferrous heme complex, we have prepared and characterized hydroperoxo-ferric complex in chloroperoxidase (CPO) and compared this to the same intermediate generated in cytochrome P450 CYP101. Optical absorption spectrum of Cpd 0 in CPO has a Soret band at 449 nm and poorly resolved alpha, beta bands at 576 and 546 nm.
Figures
Similar articles
-
Preparation and properties of ferrous chloroperoxidase complexes with dioxygen, nitric oxide, and an alkyl isocyanide. Spectroscopic dissimilarities between the oxygenated forms of chloroperoxidase and cytochrome P-450.J Biol Chem. 1985 Dec 15;260(29):15530-5. J Biol Chem. 1985. PMID: 2999120
-
Quantum mechanical/molecular mechanical study on the mechanisms of compound I formation in the catalytic cycle of chloroperoxidase: an overview on heme enzymes.J Phys Chem B. 2008 Aug 7;112(31):9490-500. doi: 10.1021/jp803010f. Epub 2008 Jul 3. J Phys Chem B. 2008. PMID: 18597525
-
Cryogenic absorption spectra of hydroperoxo-ferric heme oxygenase, the active intermediate of enzymatic heme oxygenation.FEBS Lett. 2002 Dec 4;532(1-2):203-6. doi: 10.1016/s0014-5793(02)03674-8. FEBS Lett. 2002. PMID: 12459490
-
Synthetic active site analogues of heme-thiolate proteins. Characterization and identification of intermediates of the catalytic cycles of cytochrome P450cam and chloroperoxidase.J Inorg Biochem. 2001 Feb;83(4):289-300. doi: 10.1016/s0162-0134(00)00175-6. J Inorg Biochem. 2001. PMID: 11293549 Review.
-
Heme-thiolate haloperoxidases: versatile biocatalysts with biotechnological and environmental significance.Appl Microbiol Biotechnol. 2006 Jul;71(3):276-88. doi: 10.1007/s00253-006-0417-3. Appl Microbiol Biotechnol. 2006. PMID: 16628447 Review.
Cited by
-
Electron paramagnetic resonance and electron-nuclear double resonance studies of the reactions of cryogenerated hydroperoxoferric-hemoprotein intermediates.Biochemistry. 2014 Aug 5;53(30):4894-903. doi: 10.1021/bi500296d. Epub 2014 Jul 21. Biochemistry. 2014. PMID: 25046203 Free PMC article.
-
Molecular Rationale for Partitioning between C-H and C-F Bond Activation in Heme-Dependent Tyrosine Hydroxylase.J Am Chem Soc. 2021 Mar 31;143(12):4680-4693. doi: 10.1021/jacs.1c00175. Epub 2021 Mar 18. J Am Chem Soc. 2021. PMID: 33734681 Free PMC article.
-
Heme-FeIII Superoxide, Peroxide and Hydroperoxide Thermodynamic Relationships: FeIII-O2•- Complex H-Atom Abstraction Reactivity.J Am Chem Soc. 2020 Feb 12;142(6):3104-3116. doi: 10.1021/jacs.9b12571. Epub 2020 Jan 28. J Am Chem Soc. 2020. PMID: 31913628 Free PMC article.
-
In Situ Structural Observation of a Substrate- and Peroxide-Bound High-Spin Ferric-Hydroperoxo Intermediate in the P450 Enzyme CYP121.J Am Chem Soc. 2023 Nov 22;145(46):25120-25133. doi: 10.1021/jacs.3c04991. Epub 2023 Nov 8. J Am Chem Soc. 2023. PMID: 37939223 Free PMC article.
-
Nanodiscs: A Controlled Bilayer Surface for the Study of Membrane Proteins.Annu Rev Biophys. 2018 May 20;47:107-124. doi: 10.1146/annurev-biophys-070816-033620. Epub 2018 Mar 1. Annu Rev Biophys. 2018. PMID: 29494254 Free PMC article.
References
-
- Sligar SG, Makris TM, Denisov IG. Thirty years of microbial P450 monooxygenase research: peroxo-heme intermediates--the central bus station in heme oxygenase catalysis. Biochem Biophys Res Commun. 2005;338:346–354. - PubMed
-
- Egawa T, Yoshioka S, Takahashi S, Hori H, Nagano S, Shimada H, Ishimori K, Morishima I, Suematsu M, Ishimura Y. Kinetic and spectroscopic characterization of a hydroperoxy compound in the reaction of native myoglobin with hydrogen peroxide. J Biol Chem. 2003;278:41597–41606. - PubMed
-
- Shintaku M, Matsuura K, Yoshioka S, Takahashi S, Ishimori K, Morishima I. Absence of a detectable intermediate in the compound I formation of horseradish peroxidase at ambient temperature. J Biol Chem. 2005;280:40934–40938. - PubMed
-
- Denisov IG, Makris TM, Sligar SG. Cryoradiolysis for the study of P450 reaction intermediates. Methods Enzymol. 2002;357:103–115. - PubMed
-
- Makris TM, Davydov R, Denisov IG, Hoffman BM, Sligar SG. Mechanistic enzymology of oxygen activation by the cytochromes P450. Drug Metab Rev. 2002;34:691–708. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

